Position statement
Assessing children and youth with global developmental delay and intellectual developmental disorder
Posted: Mar 27, 2026
Principal author(s)
Scott McLeod MD, Anne Kawamura MD, Angela Orsino MD, Canadian Paediatric Society, Mental Health and Developmental Disabilities Committee
Abstract
The paediatric health care provider (HCP) plays a pivotal role in the care of children with global developmental delay (GDD) and intellectual developmental disorder (IDD). This role encompasses early detection of developmental concerns, guiding families through the diagnostic process, coordinating care with other HCPs and educators, and ensuring that appropriate interventions are accessed. A comprehensive review of the medical history, completion of a thorough physical assessment, and the initiation of a genomic investigation—depending on clinical indications and available resources—are the mainstays for assessing children with GDD/IDD. Metabolic testing is only recommended in specific circumstances, which are briefly described.
Keywords: Assessment; Developmental disabilities; Genetic testing; Patient care planning
Definitions
Global developmental delay (GDD) is a neurodevelopmental disorder characterized by significant delays in achieving at least two developmental domains, including gross or fine motor, speech and language, cognitive, social, and personal functioning, and adaptive functioning[1]. A diagnosis of GDD is applied to children under the age of 5 years, who are too young to participate in formal cognitive testing (e.g., standardized intellectual tests such as the Wechsler Intelligence Scale for Children [WISC] or Standford-Binet)[1]. GDD is diagnosed based on a combination of clinical observations and structured developmental assessments. GDD has been defined variably across the literature, and there are no universally accepted or required numerical scores on developmental assessment tools that establish a diagnosis of GDD[1][2]. By contrast, intellectual developmental disorder (IDD) is characterized by limitations in both intellectual functioning and adaptive behaviour, both confirmed through standardized clinical assessment and an individual’s capacity to meet sociocultural criteria for personal independence and social responsibility[1]. Because GDD is a diagnosis reserved for children under the age of 5, HCPs must consider a formal assessment for IDD during childhood or adolescence to ensure supports continue and ensure eligibility for adult services.
IDD is often categorized by psychologists and in formal reports by its severity, which commonly reflects the level of support an individual needs or may benefit from. The DSM-5-TR also categorizes IDD by severity, but the paediatrician can usually take a more practical approach to care by focusing on individualized planning, care, and support to optimize a child or youth’s everyday function and quality of life.
Prevalence
GDD is present in an estimated 1% to 3% of children <5 years old worldwide, but its recognition and diagnosis are influenced almost everywhere by societal factors (e.g., access to early intervention programs and specialized health care resources), cultural attitudes to developmental disorders[3], and social determinants of health (e.g., food insecurity, because nutritional deficiencies can modulate presentation of GDD)[4].
The prevalence data for IDD has remained quite stable over time. A recent global estimate was 1.5% in high-income countries and 3.1% overall, suggesting a significantly higher prevalence of IDD in low- and middle-income countries[5]. However, within specific populations of individuals diagnosed with autism spectrum disorder (ASD) there has been a decline in the proportion of those with co-occurring IDD[6].
Roles of paediatric HCPs
Paediatric HCPs are well-positioned to detect developmental delays early, which is critical for improving long-term outcomes for children and youth with GDD/IDD[7]. Although one-time, cross-sectional developmental screening is not recommended for the general population[8], longitudinal developmental surveillance using tools such as the Rourke Baby Record in Canada, alongside other more targeted developmental assessments, are recommended here and elsewhere[9].
When a developmental delay is suspected, HCPs guide families through the assessment process. This includes conducting a developmental, medical, and family history. To review developmental history, HCPs can use existing resources, such as milestone charts and accessible screening tools, before referring a child to colleagues for more specific, standardized assessment. Integrating discipline-specific assessments with informed clinical understanding of each child’s developmental capacity helps determine the diagnosis of GDD. HCPs play a key role in explaining this diagnosis and the process of referrals to families, in synthesizing findings from multiple sources, and communicating these results to parents and other caregivers. Providing such guidance helps parents understand their child’s developmental profile, alleviates anxiety, and informs next steps in management and intervention.
HCPs also have an essential role in collaboration with a child or youth’s care team which, for both conditions, might include a developmental paediatrician, neurologist, geneticist, and allied health specialists. Being an effective communicator and coordinator can be pivotal for comprehensive care planning and implementation to meet child- or youth-specific needs. Also, HCPs can liaise with early childhood and school educators to ensure that each child’s or youth’s developmental needs are accommodated appropriately, while advocating for special education services and individualized education plans as needed. HCPs have a further role to play in assisting families with addressing health and behavioural concerns, and keeping them connected with community resources and support groups.
Prognoses
Prognosis for GDD is a complex and evolving field based on medical evidence and current literature. One Canadian retrospective study from 2009 found a wide distribution of cognitive outcomes in children diagnosed with GDD, with 73% scoring an IQ of 70 or above, which suggests that a substantial proportion did not meet criteria for IDD despite an earlier GDD diagnosis[10]. This study had some notable limitations but found that while fine motor and expressive language skills were significantly associated with cognitive performance, receptive language skills were not a strong predictor of later cognitive outcomes[10]. A more recent retrospective study found a stronger correlation between early developmental assessment scores and later intellectual function: children who scored ≥2 standard deviations below mean on the Griffiths Mental Development Scales at 2 to 3 years of age had an 86% likelihood of testing within the intellectual impairment range (FSIQ ≤70) on Stanford-Binet 5 (SB5) at 4 to 6 years old[11]. Such mixed evidence suggests that GDD prognosis can always benefit from domain-specific testing and long-term monitoring and follow-up for individualized care and planning.
Possible etiologies
GDD and IDD are associated with a variety of etiologies that can be broadly categorized into prenatal, perinatal, and postnatal factors as described in Table 1[12]. Note that the presence of prenatal alcohol exposure (PAE) or risk factors on history do not negate the need for comprehensive etiological evaluation or developmental assessment in children or youth with GDD/IDD.
|
Table 1. Etiologies for global developmental delay (GDD) and intellectual developmental disorder (IDD) |
|
|
Timing of exposure |
Examples |
|
Prenatal |
Infectious - Congenital infections (cytomegalovirus, herpes simplex, rubella, syphilis, toxoplasmosis, varicella, zika) Central nervous system - Holoprosencephaly, polymicrogyria Genetic/metabolic - Prader Willi syndrome, inborn errors of metabolism Toxic exposures - Ethanol, illicit substances, teratogenic medications |
|
Perinatal |
Prematurity Hypoxic ischemic encephalopathy |
|
Postnatal |
Infectious - Meningitis, encephalitis Traumatic brain injury Toxic exposures - Lead Metabolic - Hypoglycemia Neglect Nutritional deficiencies - Iron, vitamin B12 |
Information based on reference 12
An approach to in-office assessment
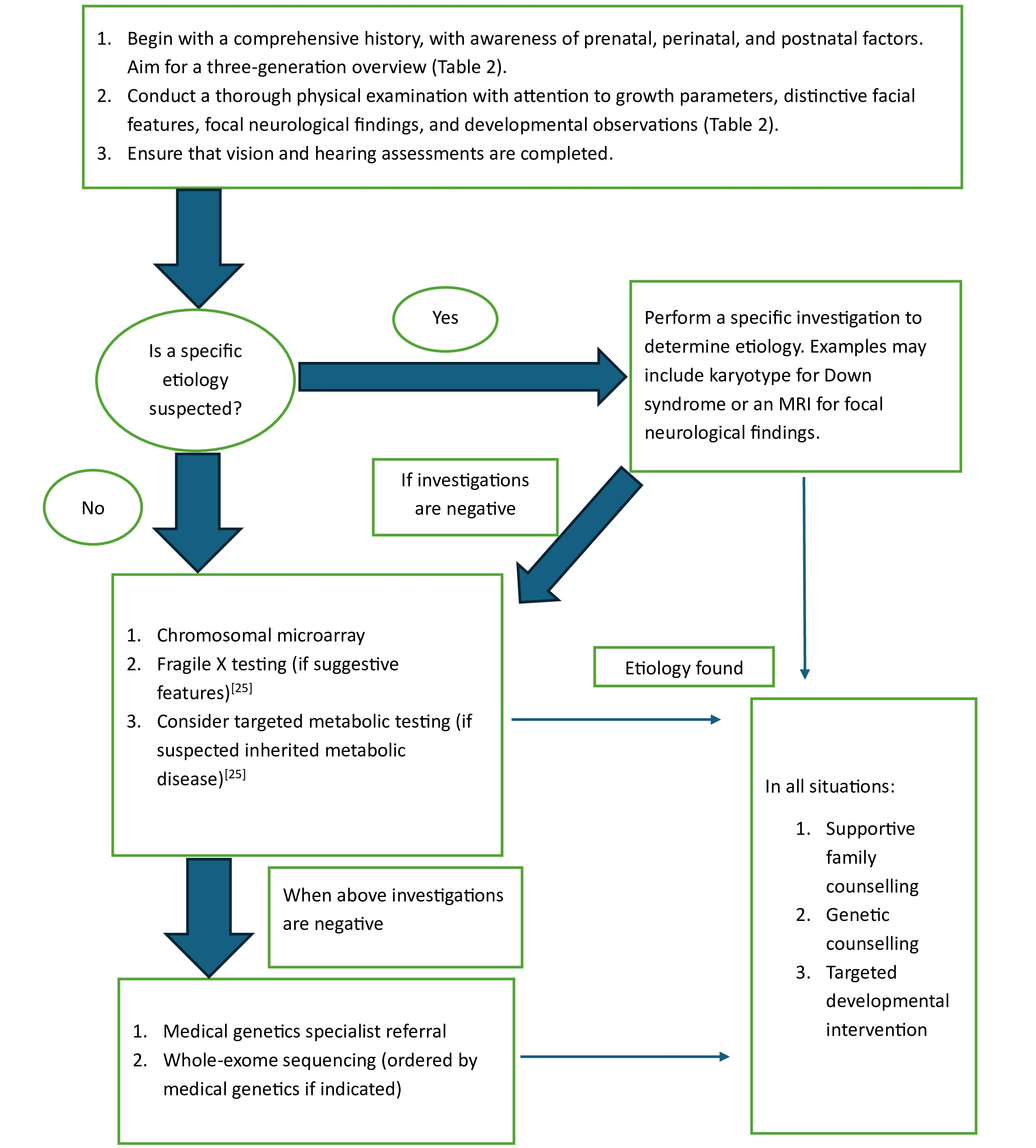
GDD and IDD present commonly in paediatrics. Initial assessment includes a focused of history and physical examination, per Table 2.
|
Table 2. History and physical examination for global developmental delay (GDD) and intellectual developmental disorder (IDD) |
|
|
HISTORY |
|
|
Concerns |
|
|
Perinatal history |
Pregnancy - maternal illnesses - exposure to medications or substances - signs of congenital infection (e.g., fever, rash) - abnormal findings on prenatal screening or ultrasound Birth history - gestational age (corrected for preterm birth) - birth weight/height/ head circumference - APGAR scores - complications during delivery and need for resuscitation Neonatal period - admission to neonatal intensive care unit - jaundice, seizures, infections (e.g., meningitis, encephalitis), metabolic issues (e.g., hypoglycemia), trauma - newborn screens, especially for newcomers to Canada |
|
Developmental review |
|
|
Family history |
Ideally, consider three generations, including:
|
|
Social history |
Enquire regarding:
|
|
Medical history |
Past:
Review:
|
|
PHYSICAL EXAMINATION |
|
|
Developmental and functional observations:
Growth parameters (height, weight, head circumference):
Head and neck:
Skin:
Cardiovascular:
Abdominal:
Neurological:
Musculoskeletal:
|
|
The HCP may need to initiate further work-up as per Figure 1.
Figure 1. Assessing global developmental delay (GDD) and intellectual developmental disorder (IDD) based on history, examinations, and a two-tiered investigation
Audiology testing
Congenital hearing loss affects 1 to 3/1000 live births and is an isolated finding in many individuals. In children with syndromic hearing loss, around 20% also have associated developmental (e.g., GDD or ASD), craniofacial (e.g., cleft palate), neurological (e.g., epilepsy), or other systemic findings (e.g., cardiac, renal, or visual impairment)[13]. Approximately 50% of infants whose deafness results from congenital hearing loss have a genetic cause, with approximately half of non-syndromic cases resulting from a gene mutation in connexine 26[13]. The evaluation of hearing in the etiological assessment of GDD/IDD is critical, especially when paired with a detailed family history over three generations. A follow-up hearing assessment is warranted to assess GDD/IDD even when the newborn hearing screen was normal[14].
Iron testing
A previous statement from the Canadian Paediatric Society proposed serum ferritin and a complete blood count as initial (tier one) laboratory investigations when evaluating for GDD/IDD[15]. A landmark longitudinal study conducted in Costa Rica found that children with chronic iron deficiency in infancy scored 8 to 9 points lower on cognitive scales than those whose early intake was normal, with continued developmental risk into later life even after treatment[16]. A recent Canadian study corroborated these results[17]. Ferritin screening is recommended for children whose families may be living with food insecurity or the following risk factors: bottle-feeding for longer, drinking more cow’s milk per day, ingesting less meat, or having a higher BMI than their peers[17]. Late identification of these children (i.e., after anemia or symptoms are already present) reduces the benefits of treatment with iron supplementation and dietary changes because chronic iron deficiency remains a developmental risk overtime.
Thyroid testing
Newborn metabolic screening can detect congenital hypothyroidism which, when untreated, can lead to impaired neurological function and growth impairment. At least two studies conducted in iodine-sufficient settings found no association between newborn thyroid-stimulating hormone (TSH) levels and cognitive scores[18][19]. TSH testing to identify etiology of GDD/IDD is likely not required as part of Canadian newborn metabolic screening. However, a TSH test may be indicated for assessing newcomer children or youth to Canada and when clinical signs suggestive of hypothyroidism are present.
Lead testing
Children can become exposed to lead by ingesting non-food items (pica). Chronic low-level exposure has been clearly associated with neurotoxicity, attentional difficulties, and lower IQ scores[20]. Active monitoring of lead levels in Canada has reduced concentrations of serum lead levels by 38% between 2007/2009 and 2018/2019[20]. Concentrations of lead are also similar between Indigenous Peoples living on reserve and the general population[20]. Routine lead testing is not recommended to evaluate GDD/IDD but may be considered when pica is present.
Neuroimaging/Neurophysiology
In a study of children and youth with GDD or IDD, brain magnetic resonance imaging (MRI) changes were found in 38% who had no neurological signs, symptoms, or established etiology for their diagnosis[21]. Abnormalities leading to an etiological diagnosis for GDD/IDD occurred in 7.9%[21]. Brain MRI findings are more prevalent in children and youth who have predominant motor delays, co-occurring visual and hearing impairments, cranial nerve abnormalities, and abnormal reflexes[22]. Neuroimaging should be considered in the presence of focal neurological findings, microcephaly, and developmental regression but should not be conducted routinely, especially because sedation may be required and unnecessary exposure to general anesthesia should be avoided.
Electroencephalography (EEG) allows for measurement of electrical activity in groups of neurons firing simultaneously. While there is a higher prevalence of EEG abnormalities in GDD/IDD, they are more strongly associated with abnormal MRI findings, genomic changes, and perinatal risk factors for GDD/IDD[23]. While EEG is useful for clarifying epilepsy and electroclinical syndromes, there is insufficient evidence to suggest it has value as a first-line investigation for either GDD or IDD.
Metabolic and genetic investigations
When a specific etiology is suspected based on clinical features, targeted genetic testing for that suspected condition should be pursued. A set of first-tier metabolic tests known as the Treatable Intellectual Disability Endeavor (TIDE) protocol was recommended to establish etiology for GDD/IDD as recently as 2018[15], although subsequent studies appear to show lower diagnostic yield from this protocol than first believed. Modifications to this testing regime[24] have since been recommended. Suggested modifications are to: 1) Only test children with GDD/IDD who have additional neurological or metabolic features (e.g., severe hypotonia, intractable seizures, developmental regression); and 2) Complete testing for plasma amino acids, total homocysteine, urine organic acids, and urine creatinine metabolites, but exclude other previously suggested tests (e.g., acylcarnitine profile, lactate, ammonia, copper, and ceruloplasmin)[24]. Metabolic investigations are no longer recommended for children or youth who do not present with neurological or metabolic features.
The Canadian College of Medical Geneticists (CCMG) released a position statement in 2023 setting out a more targeted approach to biochemical and metabolic investigations generally and a novel suggestion for Fragile X testing in particular. Rather than testing for Fragile X initially, as a first-tier diagnostic test for individuals presenting with GDD, ID, or ASD, the CCMG now recommends testing based on history and physical examination (i.e., only testing in the presence of suggestive clinical features and/or family history)[25]. The statement continues to advocate for chromosomal microarray (CMA) as a first-tier test for the genomic evaluation of a child or youth with GDD or IDD[25]. This test detects copy number variations—small duplications or deletions of genetic material—which represent alterations in the number of copies of specific genes or segments of DNA[25].
Whole exome sequencing (WES) is a next-generation sequencing (NGS) approach that analyzes the protein-coding regions (exons) of all known genes in the human genome. Because exons account for approximately 1% to 2% of the genome but contain about 85% of known disease-causing variants, WES offers a high-yield, cost-effective alternative to whole genome sequencing (WGS) for identifying genetic etiologies of neurodevelopmental disorders, including GDD and IDD[25]. The CCMG recommends WES as a second-tier genetic test for individuals with GDD and IDD, to be conducted when first-tier testing (e.g., chromosomal microarray) has not established etiology[24]. The diagnostic yield of WES for GDD/IDD, which varies between 26% and 31%, is based on findings from large clinical studies[26][27]. When testing includes WES for both biological parents as well as their child, the diagnostic yield increases compared with a proband-only approach[25]. As access to genetic counselling and testing improves over time, WES may become a first-tier test, in line with recommendations from the American Academy of Pediatrics[28]. For patient comfort, however, blood draws for genetic investigations should be minimized and secured in DNA (deoxyribonucleic acid) banks whenever possible.
Supportive care
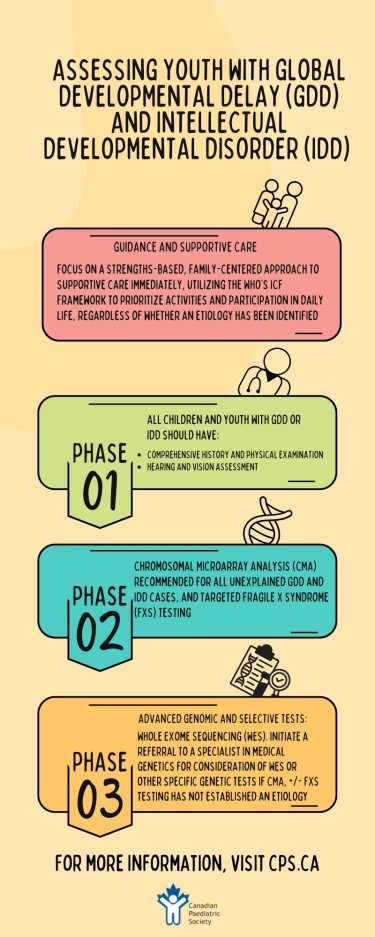
While the search for etiology can provide distinct benefits—such as enhancing caregiver understanding of the causes of developmental difference, informing health care management and surveillance, clarifying prognosis, connecting families with communities living with the same condition, and (in some cases) facilitating access to research—supporting a child’s or youth’s functional strengths and areas of need should always be a primary focus. HCPs can use the World Health Organization’s International Classification of Functioning (ICF) framework to guide practical, supportive care for individuals[29]. The ICF framework outlines body structure and functions relating to the targeted medical assessment described above and optimizing general health. However, and most importantly, the ICF focuses on the activities of daily living, which can be supported through targeted therapies such as occupational therapy to support everyday function (e.g., hygiene and dressing), speech and language therapy (including augmentative and alternative communication strategies), and emphasizes participation through recreational activities adapted to meet the needs of individuals[29]. Whenever possible, children and youth with GDD/IDD should be included in care planning and goal-setting in ways that are developmentally appropriate and aligned with capacity.
Beyond a child or youth’s intrinsic abilities, personal factors—including family values, cultural beliefs, and individual preferences—must be considered when making recommendations for supportive services and family-centred care. Families have distinct perspectives on the role of therapies, functional goals, and the acceptability of interventions.
Navigating government-supported services presents another challenge for families because programs vary widely across Canada and can be difficult to access. Complex application processes, inconsistent eligibility criteria, and jurisdictional variance create substantial barriers for families seeking assistance. Families changing jurisdictions often need to reapply for new supports, a process that can be time-consuming, burdensome, and disruptive of care continuity. HCPs should consider referring families to care navigators or social work services, when available, to facilitate access to funding, therapy programs, and school-based accommodations. Also, advocacy by care providers for service harmonization across provinces and territories actuates systemic improvement.
Conclusion
The assessment of GDD and IDD is an ongoing, evidence-based process that is adapting as new diagnostic technologies emerge. While genetic evaluation is becoming more accessible and may shift future recommendations, current best practices remain anchored in thorough history-taking, physical examination, and targeted investigations. Collaboration among HCPs facilitates research, ensures consistent medical education, and improves diagnostic efficiency. Beyond diagnosis, a strengths-based, family-centred approach is essential for supporting children and youth with GDD and IDD.
Recommendations
- A comprehensive history and physical examination, along with hearing and vision assessments, should be conducted for all children with suspected global developmental delay (GDD) or intellectual development disorder (IDD).
- In unexplained GDD/IDD, a chromosomal microarray analysis is recommended as a first-tier investigation.
- Testing for Fragile X should be considered based on clinical features, family history (or both), rather than conducted routinely for all individuals with GDD or IDD.
- After a diagnosis of GDD or IDD, clinical and educational supports should be individualized, based on domains requiring support and in line with family goals and values.
- Harmonizing government-supported and provincial/territorial programs helps ensure continuity of care for children and youth with GDD/IDD.
- Education around and use of exome sequencing as a diagnostic test for children and youth with GDD/IDD should be generously funded, based on its high diagnostic yield and clinical utility.
- Remuneration structures for paediatric health care providers (e.g., billing codes) should be updated to recognize and reimburse time spent on care coordination, communications with school and allied professionals (e.g., speech and language pathologist, occupational therapist), and completion of forms, among other duties necessary for appropriate care and advocacy for children and youth with GDD and IDD.
Acknowledgement
This statement was reviewed by the Community Paediatrics Committee and Developmental Paediatrics Section Executive of the Canadian Paediatric Society.
CANADIAN PAEDIATRIC SOCIETY MENTAL HEALTH AND DEVELOPMENTAL DISABILITIES COMMITTEE (2025-2026)
Members: Scott McLeod MD (Chair), Amy Ornstein MD (Board Representative), Natasha Saunders MD, Megan Thomas MBCHB, Ripudaman Minhas MD, Lester Liao MD, Man Ying Bernice Ho BSC (Resident Member)
Liaisons: Olivia MacLeod MD (Canadian Academy of Child and Adolescent Psychiatry), Angela Orsino MD (CPS Developmental Paediatrics Section), Leigh Wincott MD (CPS Mental Health Section)
Principal authors: Scott McLeod MD, Anne Kawamura MD, Angela Orsino MD
Funding
There is no funding to declare.
Potential Conflict of Interest
Dr. Scott McLeod reported receiving compensation for serving on the advisory board of Ipsen Biopharmaceuticals.
Other authors reported no conflicts of interest.
References
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders –TR, 5th edn. Washington, DC: APS; 2022.
- Mithyantha R, Kneen R, McCann E, Gladstone M. Current evidence-based recommendations on investigating children with global developmental delay. Arch Dis Child 2017;102(11):1071–6. doi: 10.1136/archdischild-2016-311271
- McKenzie K, Milton M, Smith G, Ouellette-Kuntz H. Systematic review of the prevalence and incidence of intellectual disabilities: Current trends and issues. Curr Dev Disord Rep 2016;3:104–15. doi: 10.1007/s40474-016-0085-7
- Bayoumi I, Parkin PC, Birken CS, Maguire JL, Borkhoff CM; TARGet Kids! Collaboration. Association of family income and risk of food insecurity with iron status in young children. JAMA Netw Open 2020;3(7):e208603. doi: 10.1001/jamanetworkopen.2020.8603
- Olusanya BO, Smythe T, Ogbo FA, Nair MKC, Scher M, Davis AC. Global prevalence of developmental disabilities in children and adolescents: A systematic umbrella review. Front Public Health 2023;11:1122009. doi: 10.3389/fpubh.2023.1122009
- Shenouda J, Barrett E, Davidow AL, et al. Prevalence and disparities in the detection of autism without intellectual disability. Pediatrics 2023;151(2): e2022056594. doi: 10.1542/peds.2022-056594
- Guralnick MJ. Early intervention for children with intellectual disabilities: An update. J Applied Res Intellect Disabil 2017;30(2):211–29. doi: 10.1111/jar.12233
- Canadian Task Force on Preventive Health Care. Recommendations on screening for developmental delay. CMAJ 2016;188(8):579–87. doi: 10.1503/cmaj.151437
- Rowan-Legg A, Bayoumi I, Kwok B, et al. The 2020 Rourke Baby Record release: A time for reflection and looking forward. Paediatr Child Health 2021;26(5):283–6. doi: 10.1093/pch/pxaa135
- Riou EM, Ghosh S, Francoeur E, Shevell MI. Global developmental delay and its relationship to cognitive skills. Dev Med Child Neurol 2009;51(8):600–6. doi: 10.1111/j.1469-8749.2008.03197.x
- Karyadiguna N, Berni C, Barnes EH, Williams K, Lo BH. Does pre-school developmental assessment agree with later intellectual assessment? A retrospective cohort study. J Paediatr Child Health 2023;59(8):962–7. doi: 10.1111/jpc.16428
- Aldosari AN, Aldosari TS. Comprehensive evaluation of the child with global developmental delays or intellectual disability. Clin Exp Pediatr 2024;67(9):435–46. doi: 10.3345/cep.2023.01697
- Gettelfinger JD, Dahl JP. Syndromic hearing loss: A brief review of common presentations and genetics. J Pediatr Genet 2018;7(1):1–8. doi: 10.1055/s-0037-1617454
- Bower C, Reilly BK, Richerson J, Hecht JL; Committee on Practice & Ambulatory Medicine, Section on Otolaryngology–Head and Neck Surgery. Hearing assessment in infants, children, and adolescents: Recommendations beyond neonatal screening. Pediatrics 2023;152(3):e2023063288. doi: 10.1542/peds.2023-063288
- Bélanger SA, Caron J. Evaluation of the child with global developmental delay and intellectual disability. Paediatr Child Health 2018;23(6):403–19. doi: 10.1093/pch/pxy093
- Lozoff B, Jimenez E, Hagen J, Mollen E, Wolf AW. Poorer behavioral and developmental outcome more than 10 years after treatment for iron deficiency in infancy. Pediatrics 2000;105(4):E51. doi: 10.1542/peds.105.4.e51
- Gingoyon A, Borkhoff CM, Koroshegyi C, et al. Chronic iron deficiency and cognitive function in early childhood. Pediatrics 2022;150(6):e2021055926. doi: 10.1542/peds.2021-055926
- Trumpff C, De Schepper J, Vanderfaeillie J, et al. No association between elevated thyroid-stimulating hormone at birth and parent-reported problem behavior at preschool age. Front Endocrinol (Lausanne) 2016;7:161. doi: 10.3389/fendo.2016.00161
- Wassie MM, Smithers LG, Yelland LN, Makrides M, Zhou SJ. Associations between newborn thyroid-stimulating hormone concentration and neurodevelopment and growth of children at 18 months. Br J Nutr 2021;126(10):1478–88. doi: 10.1017/S0007114521000325
- Health Canada. Lead in Canadians. 2021. Ottawa, Ont. December 14, 2021. Lead in Canadians (Accessed November 5, 2025).
- Murias K, Moir A, Myers KA, Liu I, Wei XC. Systematic review of MRI findings in children with developmental delay or cognitive impairment. Brain Dev 2017;39(8):644–55. doi: 10.1016/j.braindev.2017.04.006
- Alamri A, Aljadhai YI, Alrashed A, et al. Identifying clinical clues in children with global developmental delay/intellectual disability with abnormal brain magnetic resonance imaging (MRI). J Child Neurol 2021;36(6):432–9. doi: 10.1177/0883073820977330
- Lee YJ, Jo YH, Choi SH, et al. Is electroencephalography useful in children with developmental delays but without overt seizures? Ann Child Neurol 2024;32(2):105–14. doi: 10.26815/acn.2024.00444
- Vallance H, Sinclair G, Rakic B, Stockler-Ipsiroglu S. Diagnostic yield from routine metabolic screening tests in evaluation of global developmental delay and intellectual disability. Paediatr Child Health 2020;26(6):344–8. doi: 10.1093/pch/pxaa112
- Carter MT, Srour M, Au PYB, et al; Canadian College of Medical Geneticists. Genetic and metabolic investigations for neurodevelopmental disorders: Position statement of the Canadian College of Medical Geneticists (CCMG). J Med Genet 2023;60(6):523–32. doi: 10.1136/jmg-2022-108962
- Yang Y, Muzny DM, Xia F, et al. Molecular findings among patients referred for clinical whole-exome sequencing. JAMA 2014;312(18):1870–9. doi: 10.1001/jama.2014.14601
- Retterer K, Juusola J, Cho MT, et al. Clinical application of whole-exome sequencing across clinical indications. Genet Med 2016;18(7):696–704. doi: 10.1038/gim.2015.148
- Rodan LH, Stoler J, Chen E, Geleske T; Council on Genetics. Genetic evaluation of the child with intellectual disability or global developmental delay: Clinical report. Pediatrics 2025;156(1):e2025072219. doi: 10.1542/peds.2025-072219
- World Health Organization. International classification of functioning, disability and health (ICF). Geneva, Switzerland: WHO; 2025 (Accessed November 5, 2025).
Disclaimer: The recommendations in this position statement do not indicate an exclusive course of treatment or procedure to be followed. Variations, taking into account individual circumstances, may be appropriate. Internet addresses are current at time of publication.