Practice point
Management of antenatal urinary tract dilation—A clinical practice point for neonatal care providers
Posted: Jan 19, 2026
Principal author(s)
Laura Betcherman MD, Charushree Prasad MD, Matt Harding MD, Robert L. Myette MD PhD, Peter Wong MD, Priya Saini MD; Canadian Paediatric Society, Community Paediatrics Committee
Abstract
Antenatal urinary tract dilation (UTD) is a common clinical scenario for paediatricians. Differing guidelines, practice variability, and inconsistent reporting of ultrasound findings make it challenging to identify infants requiring follow-up, time postnatal imaging, and determine the need for antibiotic prophylaxis. Both antenatal and postnatal ultrasounds should assess the anterior–posterior renal pelvic diameter (APD) and for the presence of any complex features. Indication for additional scans, prophylactic antibiotics, and subspecialty involvement are based on these findings. The evidence for use of prophylactic antibiotics is limited but may be considered in a shared decision-making model with the family for patients with complex features or increased APD measurement. This practice point integrates existing guidelines and best evidence to provide a clinical pathway for managing antenatal UTD.
Keywords: Antenatal hydronephrosis; Prophylactic antibiotics; Uretero-pelvic junction obstruction; Urinary tract dilation; Vesicoureteric reflux
Background
Urinary tract dilation (UTD) is a commonly detected abnormality on prenatal ultrasound (US), seen in up to 5% of pregnancies. Although most cases are transient, UTD has been associated with congenital anomalies of the kidney and urinary tract[1][2]. These conditions include vesicoureteric reflux (VUR; 10% to 40%), uretero-pelvic junction obstruction (UPJO; 10% to 30%), uretero-vesical junction obstruction (UVJO; 5% to 10%), primary megaureter (5% to 15%), and posterior urethral valves (PUV; 1% to 5%)[3]. Early recognition and management of UTD is important to prevent complications such as recurrent urinary tract infections (UTIs), nephrolithiasis, and obstructive uropathy, all of which can lead to chronic kidney disease (CKD)[2]. More severe antenatal UTD is correlated with higher risk for clinically significant pathology[4][5].
Definitions of antenatal hydronephrosis can be vague due to widely variable descriptive terminologies. For this practice point, UTD is used instead of antenatal hydronephrosis, as a more inclusive term. Traditionally, intra-renal anterior–posterior pelvic diameter (APD) has been used to stratify UTD, but this measurement can fluctuate somewhat with physiological changes, fluid status, US probe positioning, and operator technique[6]. To determine the significance of the UTD, APD should be assessed alongside any complex features (Table 1)[7][8]. Note that dilation of the extra-renal pelvis is not associated with renal parenchymal thinning and therefore is not considered a complex feature.
| Table 1. Complex features associated with urinary tract dilation (UTD) | ||
| Upper urinary tract | ||
| Bilateral UTD |
|
|
| Solitary kidney with UTD |
|
|
|
Dysplastic kidney(s) with UTD |
|
|
|
Peripheral calyceal dilation |
|
|
| Parenchymal thinning |
|
|
| Lower urinary tract | ||
|
Bladder abnormality |
|
|
|
Ureter abnormality |
|
|
| Fetus | ||
|
Amniotic fluid abnormality
|
|
|
APD, anterior–posterior renal pelvic diameter; CKD, chronic kidney disease; PUV, posterior urethral valves
Overview of existing guidelines
Challenges exist in caring for infants with antenatal UTD because there is no definitive resource for management and postnatal follow-up is highly variable. Two main guidance documents that consider both APD and complex features are the Canadian Urological Association (CUA) guideline and the multidisciplinary UTD classification system[9][10]. Both show good inter- and intra-reliability in reporting UTD[11]. The CUA defines antenatal UTD using the existing Society of Fetal Urology (SFU) classification, and considers both APD and presence of cortical thinning[9]. The updated UTD classification system focuses on defining abnormalities seen antenatally (A) and postnatally (P) and assigning grades of severity[8][10]. Higher risk categories (A2-3, P2-3) are assigned based on increased APD along with abnormalities in the calyces, ureter, bladder, and renal parenchyma[8][10]. While both classification systems can aid in postnatal UTD management, there are inconsistencies between the two in reporting complex features radiographically. Discrepancies can lead to difficulties with interpretation and communication among providers and when analyzing patients’ surgical and research outcomes[8][9].
A new guideline was published by the American Academy of Pediatrics (AAP) in 2025, with recommendations adapted from the UTD classification system that generally align with this CPS practice point[12]. Specifically, both documents focus on limiting antibiotics and tailoring individual management in accordance with the presence or absence of qualifying additional features and AP diameter when treating children with UTD.
Prophylactic antibiotics
Continuous antibiotic prophylaxis (CAP) has been used to reduce the incidence of UTIs in children with UTD. The underlying principle of this treatment is that urinary stasis and reflux are less likely to cause UTIs if urine is sterile[13]. Disadvantages of CAP include side effects, cost, pill burden, and risk for antibiotic resistance. Recommendations are based largely on expert consensus and to date there is no strong evidence to support CAP.
A 2013 systematic review by the CUA showed no benefit from CAP in low-grade hydronephrosis (SFU 1-2), but in higher grade (SFU 3-4) there was a significant reduction in UTI rates in infants on CAP (14.6%) compared with those not on CAP (28.9%, P<0.01)[14]. A subsequent review in 2017 found no significant difference in UTI reduction across all grades of hydronephrosis[13]. In both studies, however, important variables such as gender, reflux, and circumcision status were not effectively analyzed[13][14] potentially limiting applicability for high-risk groups.
A contemporaneous systematic review by the European Society for Paediatric Urology found insufficient evidence to support routine use of CAP in neonates with UTD, but in specific sub-groups (uncircumcised males, hydroureteronephrosis, high-grade UTD) there was potential for benefit[15]. A 2023 randomized controlled trial (RCT) on infants with grade 3-5 VUR and no prior UTI concluded that routine use of CAP is not justified but that CAP use in female patients with grade 4-5 VUR may be beneficial[16]. In this study, CAP was associated with both an increased incidence of non-E. coli-associated UTIs and increased antibiotic resistance (52% versus 17%)[16].
Reducing UTI burden and sequelae are usually cited as reasons for CAP. However, one systematic review showed no influence of CAP in preventing renal scarring in children with recurrent UTIs, although the authors were unable to comment on utility in patients with high-grade reflux, renal dysplasia, or both[17].
Overall, because the data fail to show significant benefit of CAP while increasing concern for antibiotic resistance, the current evidence does not support routine CAP use to treat UTD. For mild presentations (e.g., APD ≤14 mm without complex features), parents should be educated around the symptoms of a UTI and when to seek medical attention. More data are needed to manage infants with severe UTD, with or without additional risk factors for UTI. For high-risk patients (e.g., APD ≥15 mm or with complex features), there should be shared decision-making with the family regarding treatment with CAP, with subspecialist involvement. Patients with suspected UPJO may not require CAP, but starting prophylaxis for high-grade UTD is reasonable while awaiting subspecialty consultation.
The most common agent used for CAP is trimethoprim (without sulfamethoxazole) at a recommended dose of 2 mg/kg/day[15]. Alternative agents include cephalexin and nitrofurantoin. Trimethoprim-sulfamethoxazole in newborns less than 2 months old increases risk for jaundice[9]. Nitrofurantoin in neonates under 1 month old may cause hemolytic anemia[9]. Local antibiograms should be consulted when determining an appropriate agent.
Clinical pathway
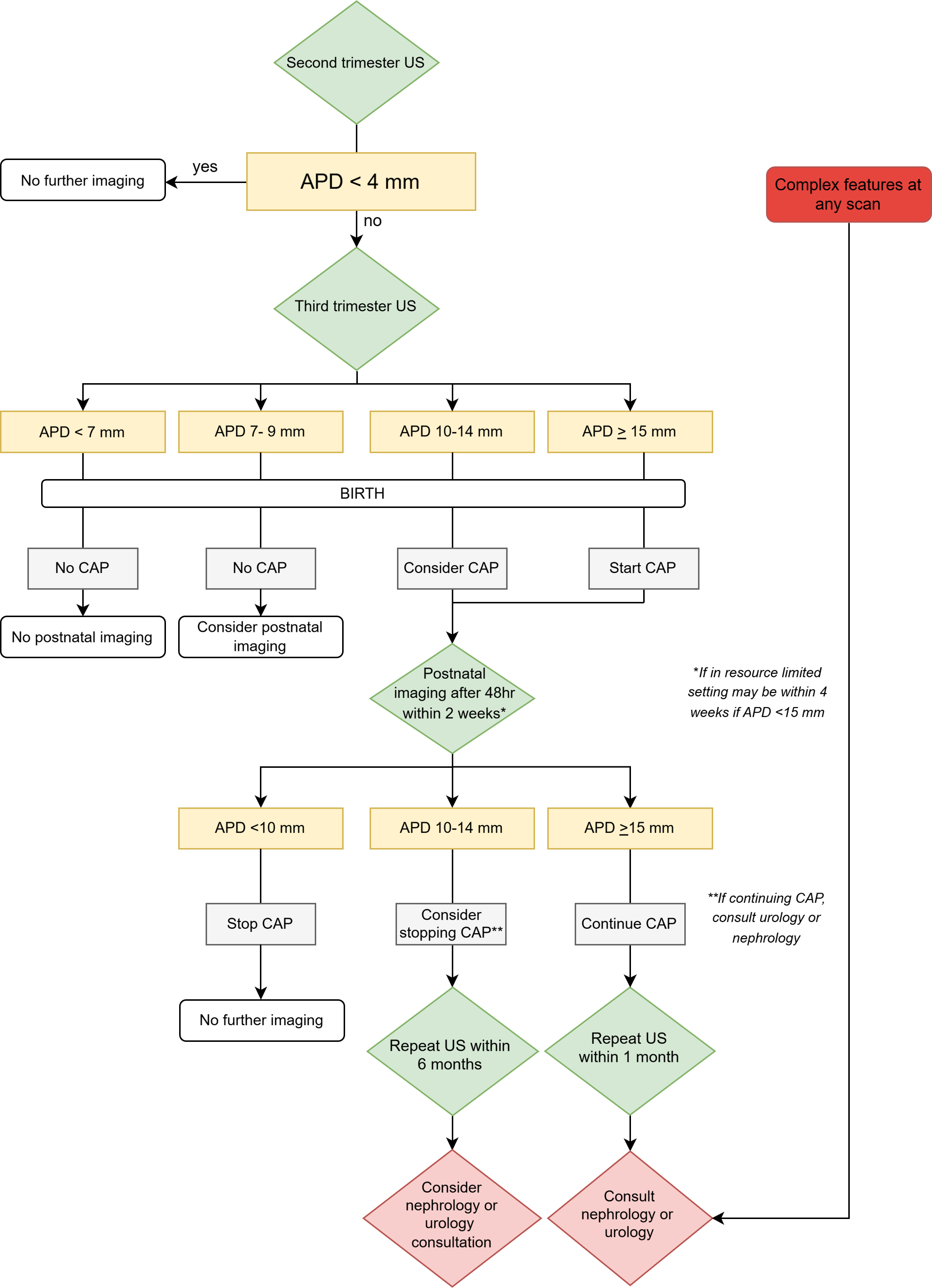
The following pathway is recommended based on expert consensus and using the UTD classification system as a reference to account for both intrarenal APD and complex features (Figure 1). Providers should counsel all families on the signs and symptoms of UTI in infants and when to seek medical attention. This pathway outlines the need for postnatal US follow-up for patients with antenatal UTD. While the risk of uropathology is low when the fetal APD is 7 to 9 mm in the third trimester, some guidelines suggest a follow-up postnatal US for these infants[9][10]. Follow-up can be considered based on access to resources, and local practice.
Figure 1. Managing antenatal urinary tract dilation (UTD)
Note: Complex features include ureter abnormality, bladder abnormality, peripheral calyceal dilation, oligohydramnios, parenchymal thinning, dysplastic or echogenic kidney(s), bilateral UTD, UTD in a solitary kidney.
APD, anterior-posterior renal pelvic diameter; CAP, continuous antibiotic prophylaxis; US, ultrasound
Timing of follow-up imaging
A US should usually be performed at least 48 hours post-birth to avoid underestimating the UTD during a state of relative volume depletion. Optimally, the US occurs within the first 2 weeks post-birth. When access to US is limited, the first imaging can be completed within one month post-birth provided the third trimester APD is <15 mm and there are no complex features.
In the absence of complex features, when the APD has remained 10 to 14 mm postnatally, the infant may be followed by a general practitioner with a US completed within 6 months. When the postnatal APD is ≥15 mm, a repeat US should be done within one month, and additional imaging (i.e., a voiding cystourethrogram or Lasix renal scan) can be considered alongside the urology/nephrology consult. If at any point the presence or absence of a complex feature has not been mentioned in the US report, and may alter the management, the provider can request a specific comment from radiology.
Postnatal imaging for antenatally suspected PUV (bilateral severe hydroureteronephrosis +/- bladder anomalies, renal parenchymal changes, oligohydramnios, or presence of a “keyhole sign”) should be completed urgently in consultation with paediatric urology. Witnessed postnatal voids do not preclude a diagnosis of severe obstruction[7].
Indications for nephrology/urology involvement
The specific indications for urology/nephrology involvement are: complex features on any imaging, persistent or worsening postnatal UTD, maintaining postnatal CAP, and/or a postnatal APD >15 mm.
Indications for blood work
Electrolytes and renal function investigations are not indicated for uncomplicated UTD, but should be ordered for infants with significant bilateral UTD suggesting renal parenchymal compression or concerns for PUV. Blood work should also be completed for any infant with bilateral kidney disease (e.g., bilateral cysts, increased echogenicity, or dysplasia).
Best practice points
- It is important to identify newborns who require urinary tract dilation (UTD) follow-up to prevent complications.
- Figure 1 guides timing of postnatal imaging for infants whose antenatal US shows UTD with an anterior-posterior renal pelvic diameter (APD) >10 mm, complex features, or both.
- More concrete guidance regarding continuous antibiotic prophylaxis (CAP) is needed, particularly for children with higher grade UTD. A shared decision-making model with the family should be employed in discussing CAP.
- Clinicians should engage with radiologists to obtain information about complex features if they were not included in the original US report.
- Referral to paediatric nephrology/urology (or both) can be made in any case of UTD (including by maternal health care providers), but should especially be made in cases with complex features.
Acknowledgement
The authors wish to thank Dr. Kourosh Afshar (urologist, BC Children’s Hospital, University of British Columbia), and the Canadian Association of Paediatric Nephrologists (CAPN) for their input and feedback. The pathway was assessed and modified by members of CAPN and the Nephrology Special Interest Group of the Canadian Pediatric Society. This practice point was reviewed by the Fetus and Newborn and Infectious Diseases and Immunization Committees of the Canadian Paediatric Society.
CANADIAN PAEDIATRIC SOCIETY COMMUNITY PAEDIATRICS COMMITTEE (2023-2024)
Members: Peter Wong MD (Chair), Jill Borland Starkes MD (Board Representative), Audrey Lafontaine MD, Michael Hill MD, Meta van den Heuvel MD, Kelcie Lahey MD MSC
Liaison: Richa Agnihotri MD (Community Paediatrics Section)
Principal authors: Laura Betcherman MD, Charushree Prasad MD, Matt Harding MD, Robert L. Myette MD PhD, Peter Wong MD, Priya Saini MD
Funding
There is no funding to declare.
Potential Conflict of Interest
The authors have indicated they have no conflicts of interest.
References
- Melo FF, Mak RH, Simões E Silva AC, et al. Evaluation of urinary tract dilation classification system for prediction of long-term outcomes in isolated antenatal hydronephrosis: A cohort study. J Urol 2021;206(4):1022-30. doi: 10.1097/JU.0000000000001899
- Lee RS, Cendron M, Kinnamon DD, Nguyen HT. Antenatal hydronephrosis as a predictor of postnatal outcome: A meta-analysis. Pediatrics 2006;118(2):586-93. doi: 10.1542/peds.2006-0120
- Vincent K, Murphy HJ, Twombley KE. Urinary tract dilation in the fetus and neonate. Neoreviews 2022;23(3):e159-e174. doi: 10.1542/neo.23-3-e159
- Rickard M, Dos Santos J, Keunen J, Lorenzo AJ. Prenatal hydronephrosis: Bridging pre- and postnatal management. Prenat Diagn 2022;42(9):1081-93. doi: 10.1002/pd.6114
- Hwang J, Kim PH, Yoon HM, et al. Application of the postnatal urinary tract dilation classification system to predict the need for surgical intervention among neonates and young infants. Ultrasonography 2023;42(1):136-46. doi: 10.14366/usg.22035
- Dos Santos JR, Zu’bi F, Hojjat A, et al. The impact of the state of hydration on the degree of hydronephrosis in children: Towards a uniform standard of ultrasonographic assessment. Pediatrics 2018;141 (1_MeetingAbstract):283.
- Coquillette M, Lee RS, Pagni SE, Cataltepe S, Stein DR. Renal outcomes of neonates with early presentation of posterior urethral valves: A 10-year single center experience. J Perinatol 2020;40(1):112-7. doi: 10.1038/s41372-019-0489-4
- Nguyen HT, Phelps A, Coley B, Darge K, Rhee A, Chow JS. 2021 update on the urinary tract dilation (UTD) classification system: Clarifications, review of the literature, and practical suggestions. Pediatr Radiol 2022;52(4):740-51. doi: 10.1007/s00247-021-05263-w
- Capolicchio JP, Braga LH, Szymanski KM. Canadian Urological Association/Pediatric Urologists of Canada guideline on the investigation and management of antenatally detected hydronephrosis. Can Urol Assoc J 2018;12(4):85–92. doi: 10.5489/cuaj.5094
- Nguyen HT, Benson CB, Bromley B, et al. Multidisciplinary consensus on the classification of prenatal and postnatal urinary tract dilation (UTD classification system). J Pediatr Urol 2014;10(6):982–98. doi: 10.1016/j.jpurol.2014.10.002
- Rickard M, Easterbrook B, Kim S, et al. Six of one, half a dozen of the other: A measure of multidisciplinary inter/intra-rater reliability of the Society for Fetal Urology and urinary tract dilation grading systems for hydronephrosis. J Pediatr Urol 2017;13(1):80.e1-80.e5. doi: 10.1016/j.jpurol.2016.09.005
- Herndon CDA, Otero HJ, Hains D, Sweeney RM, Lockwood GM, AAP Sections on Urology, Nephrology, Radiology and Hospital Medicine. Perinatal urinary tract dilation: Recommendations on pre-/postnatal imaging, prophylactic antibiotics, and follow-up; Clinical report. Pediatrics 2025;156(1):e2025071814. doi: 10.1542/peds.2025-071814
- Easterbrook B, Capolicchio JP, Braga LH. Antibiotic prophylaxis for prevention of urinary tract infections in prenatal hydronephrosis: An updated systematic review. Can Urol Assoc J 2017;11(1-2Suppl1):S3-S11. doi: 10.5489/cuaj.4384
- Braga LH, Mijovic H, Farrokhyar F, Pemberton J, DeMaria J, Lorenzo AJ. Antibiotic prophylaxis for urinary tract infections in antenatal hydronephrosis. Pediatrics 2013;131(1):e251-61. doi: 10.1541/peds.2012-1870
- Silay MS, Undre S, Nambiar AK, et al. Role of antibiotic prophylaxis in antenatal hydronephrosis: A systematic review from the European Association of Urology/European Society for Paediatric Urology Guidelines Panel. J Pediatr Urol 2017;13(3):306-15. doi: 10.1016/j.jpurol.2017.02.023
- Morello W, Baskin E, Jankauskiene A, et al; PREDICT Study Group. Antibiotic prophylaxis in infants with grade III, IV, or V vesicoureteral reflux. N Engl J Med 2023;389(11):987-97. doi: 10.1056/NEJMoa2300161
- Hewitt IK, Pennesi M, Morello W, Ronfani L, Montini G. Antibiotic prophylaxis for urinary tract infection–related renal scarring: A systematic review. Pediatrics 2017;139(5):e20163145. doi: 10.1542/peds.2016-3145
Disclaimer: The recommendations in this position statement do not indicate an exclusive course of treatment or procedure to be followed. Variations, taking into account individual circumstances, may be appropriate. Internet addresses are current at time of publication.