Position statement
Premedication for laryngoscopy in the newborn infant
Posted: Apr 20, 2026
Principal author(s)
Anne-Sophie Gervais MD, Keith J Barrington MB, Emer Finan MB; Canadian Paediatric Society, Fetus and Newborn Committee
Abstract
Laryngoscopy is commonly performed in the neonatal intensive care unit and is known to be associated with adverse effects. The use of premedication has been shown to decrease these effects and increase success rates. In all non-emergent intubations, premedication with a vagolytic, fast-acting opiate and short-acting muscle relaxant should be provided. For less invasive surfactant administration, non-pharmacological measures should always be prioritized. Fast-acting opiate agents are also recommended combined with a vagolytic agent. Laryngoscopy should always be performed or supervised by a skilled provider, with constant cardiorespiratory monitoring in place.
Keywords: Laryngoscopy; Less invasive surfactant administration; Minimally invasive surfactant therapy; Newborn; Premedication; Surfactant administration
Laryngoscopy is one of the most common procedures performed in neonatal intensive care units (NICUs) and is recognized to be a painful and stressful experience[1][2]. The past many years have seen increasing use of premedication for laryngoscopy, with significant variation in practice[3][4]. More recent techniques for less or minimally invasive surfactant administration (LISA or MIST) have been developed[5], which also require laryngoscopy. Lack of consensus remains regarding premedication use[6]-[9] and preferred agents for these newer techniques for surfactant administration.
The effects of untreated pain and stress in neonates are increasingly recognized, alongside growing evidence that repeated exposure to pain and stress has adverse effects[1][10]-[12] and long-term neurodevelopmental impacts on the developing brain[13]. It is also an ethical duty of physicians and other caregivers to minimize pain and use analgesia for non-emergent painful procedures.
This statement replaces Canadian Paediatric Society guidance from 2011[14] and provides recommendations on premedication for both laryngoscopy and newer surfactant administration techniques.
What are the physiological responses to laryngoscopy?
Most research in this area has focused on the physiological responses to laryngoscopy when accompanied by endotracheal intubation, but laryngoscopy can have major impacts even before passage of the endotracheal tube. Physiological responses to awake laryngoscopy in neonates include changes in heart rate, blood pressure, oxygen saturation, and intracranial pressure[15]. An inadequately sedated infant who is resisting the procedure can experience an increase in intrathoracic pressure with decreased venous return, causing intracranial hypertension with potential for intraventricular hemorrhage[16] and periventricular leukomalacia[17][18]. Increases in intracranial pressure can also be caused by hypoxia or hypercapnia and extension of the neck causing venous obstruction[19]. These effects can all be counteracted by premedication with neuromuscular relaxants, which reduce or eliminate responses that increase intracranial pressure[20]. Laryngoscopy and endotracheal intubation cause arterial pressure to rise dramatically, often to a >60% increase in mean blood pressure (BP)[17][20]. Hypoxemia is likely due, in part, to apnea, which may result from vagal stimulation. Also, distortion of the pharynx and upper airway can occur during laryngoscopy, leading to airway obstruction that further contributes to hypoxemia[21]. Laryngoscopy frequently causes a decrease in heart rate, likely due to vagal stimulation and prolonged procedural attempts causing hypoxemia[22].
Should premedication be used before laryngoscopy?
Premedication with diverse agents (vagolytics, analgesia, sedation, muscle relaxants) has beneficial effects, including increased procedural success, shorter duration of attempts, lower pain and stress, and fewer adverse physiological effects[1][2][15][23]-[26]. Limiting both the number of attempts and duration of each is crucial to minimize associated physiological disturbances[21]. Another key step toward optimizing physiological conditions is to stabilize neonates before the procedure, including ensuring adequate oxygenation, which limits risk for procedural deterioration. Optimal respiratory support and targeting normal saturations pre-procedure are especially important[27].
What medications should be used before laryngoscopy?
Unless there is a significant reason for avoiding medication, appropriate analgesia should be given to neonates before non-emergent laryngoscopy attempts[2][13][28]. Moreover, administering an anticholinergic (atropine or glycopyrrolate) in addition is effective to prevent bradycardia[29][30]. The characteristics of various drugs that have been studied for premedication use in neonatal intubation are outlined below.
Opiates
Opioids are often used for neonatal pain management and provide analgesia and varying degrees of sedation. Morphine has been well studied for these uses but is ineffective in reducing physiological instability (e.g., hypoxia, bradycardia, systemic hypertension) or the time needed to perform intubation, likely due to its delayed onset of action[17]. Fentanyl has a more rapid onset of action (2 to 3 minutes), and is more effective than morphine for reducing pain and decreasing instability. Remifentanil is also rapidly effective and has the added advantage of being cleared quickly[24]. However, both fentanyl and remifentanil carry risk for causing chest wall rigidity[16], and may also cause hypoventilation. They should be given by slow intravenous (IV) push over 2 to 5 minutes, including the first portion of the flush, and the care team must be ready to provide assisted ventilation in case of apnea, hypoventilation, or chest wall rigidity. The use of muscle relaxants can reverse chest wall rigidity[22][31]. The use of intranasal (IN) fentanyl in neonates has also been reported for a range of neonatal procedures, including intubation[32]-[34], but its onset of action is prolonged and use should be restricted to scenarios where venous access is not possible.
Propofol
Propofol is a general anesthetic that is widely used for intubation in adults. One randomized trial (RCT) of 63 infants comparing propofol with a combination of morphine, atropine, and succinylcholine for intubation showed that infants who received propofol had a successful intubation twice as fast compared with the morphine, atropine, and succinylcholine group, without significant difference in heart rates and blood pressures[35]. Another comparison of propofol and atropine with atracurium, sufentanil, and atropine showed better sedation and more rapid intubation in the sufentanil group[36]. Propofol has been shown to cause hypotension however, especially at the high levels required to achieve adequate sedation. Therefore, propofol is not commonly used in neonates[37]-[39].
Benzodiazepines
Use of benzodiazepines before a procedure provides both sedation and amnesia, but no analgesic effect. Therefore, benzodiazepines should not be used alone, and there are no reliable trial data showing clinical benefit from adding midazolam to an analgesic. Midazolam is the most commonly used benzodiazepine and is often combined with an opioid[15]. Midazolam has a fast onset of action and can also be administered IN (with a much slower onset of action) when an infant does not have an IV access[15][16]. Midazolam has a hypotensive effect and a long and variable half-life, which can delay recovery of spontaneous breathing[40]. Midazolam is also associated with a higher rate of adverse events, especially when used as a sedative long term or in more preterm infants[41][42].
Ketamine
The evidence regarding ketamine use for intubation in neonates is scarce. The only randomized, controlled study to date in neonates (n=60) compared IN ketamine with IN midazolam and showed less effective sedation with ketamine than with midazolam. Similar hemodynamic and respiratory effects were noted between the two drugs[43].
Anticholinergics/vagolytics
Atropine is an anticholinergic medication used to prevent reflex bradycardia due to increased vagal response during laryngoscopy and intubation[16]. Atropine may also reduce secretions before this procedure[44]. Glycopyrrolate is an atropine analogue that does not cross the blood–brain barrier and has also been shown to prevent bradycardia during intubation[45].
Muscle relaxants
Succinylcholine is a depolarizing muscle relaxant with a very rapid onset of action (1 minute) and short duration (10 minutes). Adverse effects of succinylcholine include bradycardia, which can be avoided by administering atropine prior to administration of a muscle relaxant[16]. Succinylcholine also carries increased risk for hyperkalemia and can trigger malignant hyperthermia[2], and should not be used in infants with hyperkalemia or a family history of malignant hyperthermia. Furthermore, succinylcholine should not be used in any child with a neuromuscular disorder. Succinylcholine has been known to abort chest wall rigidity caused by opioid administration.
Non-depolarizing muscle relaxants include rocuronium, pancuronium, and atracurium. Rocuronium has short onset of action (1 to 3 minutes) but a longer and variable duration of action (of up to an hour), which makes it less appropriate for intubation than succinylcholine[14][15]. Pancuronium was the first muscle relaxant studied for this purpose but is no longer available due to risk for tachycardia. Another negative effect is its prolonged duration of action. Atracurium was studied as one component of combined premedications in a single multicentre RCT (n=173), which showed that return of spontaneous movement and respiration took more than 30 minutes (median) while the 75th percentile took almost one hour for recovery of respiration and 65 minutes to recovery of spontaneous limb movement[36].
What are the complications of premedicating an infant for intubation?
Short-term side effects (e.g., hemodynamic instability) have been described with pre-medication, along with unclear effects on long-term outcomes. Some studies have shown a drop in BP without attendant impacts on cerebral oxygenation or cardiac output[46][47]. There is no evidence of significant harm associated with side effects of premedication for intubation. There is also no evidence that administering occasional doses of opiates has a negative impact on neurological outcomes in neonates[4], while there is ample evidence that repeated, painful procedures are associated with worse outcomes[2][10]-[12]. Multiple attempts at intubation are also associated with higher rates of intraventricular hemorrhage[48]. The number of procedural attempts can be reduced with appropriate premedication.
When is it acceptable to intubate an infant without premedication?
Premedication is recommended for all non-urgent intubations. Recent Italian guidelines[2] made this a strong recommendation based on evidence of moderate quality, while one French review[15] also strongly recommended using premedication beyond “immediate life-threatening situations”. Preponderance of evidence supports administering premedication unless an urgent clinical need prevents safely doing so (e.g., in a neonatal resuscitation setting).
In scenarios where IV access cannot readily be obtained, administering IN fentanyl, which has an onset of action of 5 to 8 minutes, is an option[49]. Every attempt should be made to prioritize IV access, however, which may be critical for managing complications. Other IN drugs, such as midazolam, have no analgesic effect, and can cause hypotension[50].
For newborns with airway anomalies, an individualized premedication regimen is needed, although analgesia should be provided.
What are the characteristics of an acceptable premedication protocol?
The benefits of combining opioid analgesia with a muscle relaxant are well established in the literature. Fentanyl or remifentanil are preferred based on their rapid onset of action, although users must be aware of and watch for signs of chest wall rigidity[51]-[53]. They must also make sure this opiate medication is administered slowly[25][52].
Recent single centre data looking at 352 intubations showed that “full premedication”, which included a vagolytic in addition to an opiate and muscle relaxant, was associated with fewer adverse events and greater success than “partial premedication” with these medications, or with no premedication at all[26]. Data from the Near4Neos multicentre cohort study examining over 2000 intubations showed a reduction in adverse events when muscle relaxation was used[25].
Propofol alone cannot be recommended for most neonatal intubations because doses high enough to blunt physiological responses often lead to hypotension[37]-[39]. Midazolam should not be used alone and has no proven benefit in this situation. There is inadequate experience with ketamine to recommend its use.
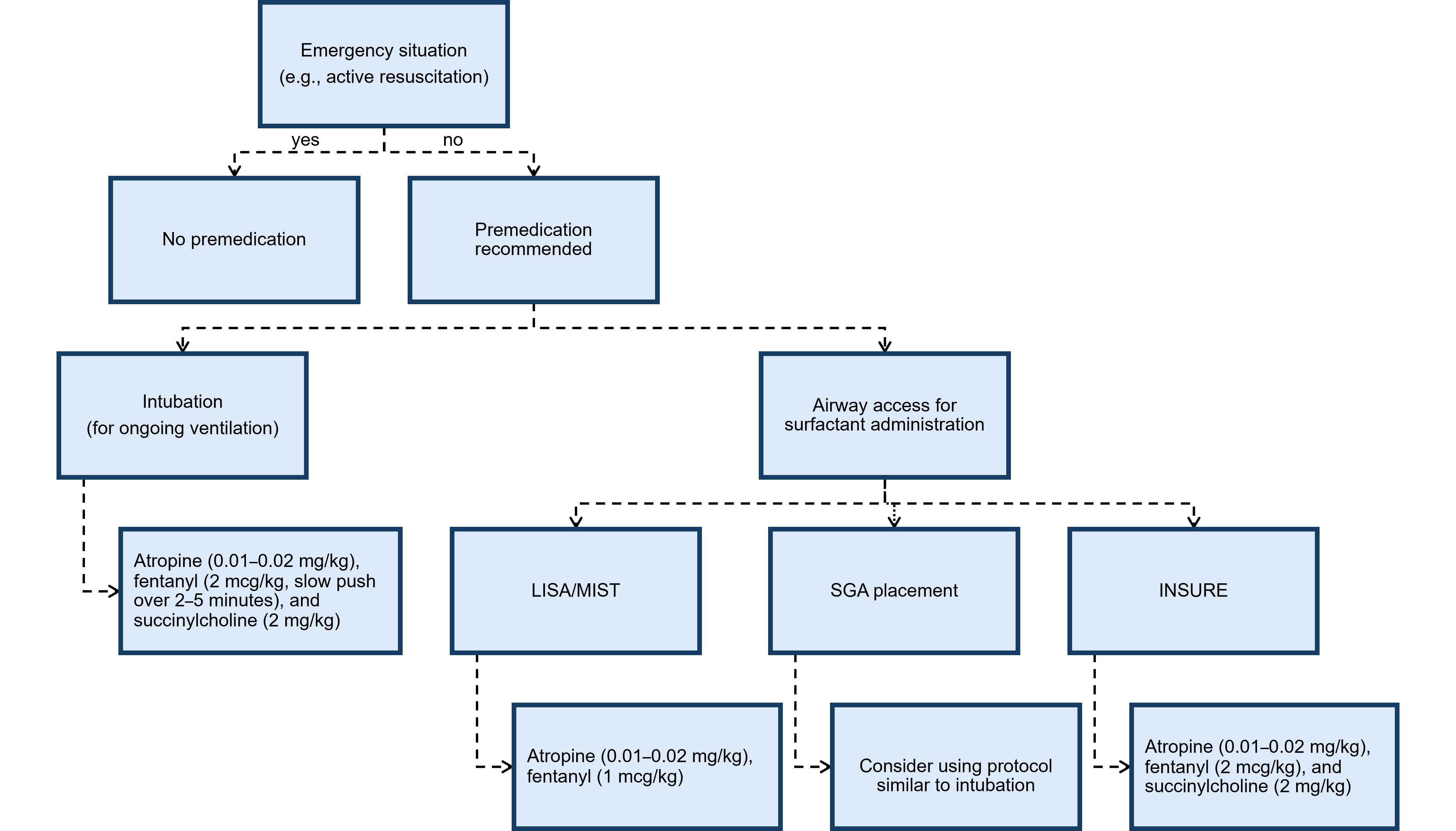
Based on available evidence, the preferred approach to premedication for intubation would be to use a vagolytic, a short-acting opioid, and a short-acting muscle relaxant. Suggested drugs and dosages are atropine (0.01 to 0.02 mg/kg), fentanyl 2 mcg/kg (administered slowly over 2 to 5 minutes), and succinylcholine (2 mg/kg)[2] (see Table 1). Remifentanil (1 mcg/kg), with its shorter duration of onset and action, may be considered as an alternative opiate.
Special considerations depending on disease process
Providers should be aware of the potential side effects and pharmacokinetic profiles of the drugs used. It should also be noted that hepatic and renal dysfunction can impact the clearance of various drugs[2]. Therapeutic hypothermia alters the effectiveness of the cytochrome 450 pathway, making clearance of opiates and midazolam less efficient. Lower dosages of both drugs may be required[2]. Remifentanil is rapidly broken down in blood and tissues and is less impacted by renal and hepatic dysfunction[54].
Premedication for surfactant administration
Recent years have seen a rapid increase in the use of non-invasive respiratory support along with the development of various techniques for less invasive methods of surfactant administration[8][9]. When an infant is not expected to remain intubated, the need to maintain or rapidly recover respiratory drive will influence the choice of premedication.
INSURE
The INSURE procedure (intubate, surfactant, extubate) administers surfactant with very rapid weaning and extubation. Drugs that lead to prolonged respiratory depression are therefore contraindicated. Use of medications such as naloxone to reverse the sedative effects of opiates and facilitate rapid extubation has also been described[55]. A vagolytic and a short-acting opiate (fentanyl/ remifentanil) and muscle relaxant (such as succinylcholine) are recommended. Newborns can have different responses to medications, however, and assessing effectiveness of respiration before extubation is essential[2].
LISA/MIST
Newer techniques of surfactant administration such as less invasive surfactant administration (LISA) and minimally invasive surfactant therapy (MIST) are increasingly used to avoid even short periods of mechanical ventilation. An infant’s respiratory drive must be maintained during either procedure, which has caused debate regarding premedication[6]-[9]. Some advocate for premedication for all, while others support an individualized approach, integrating non-pharmacological measures first, then using pharmacological measures as required[9]. A cross-Canada survey published in 2022 showed that in the 17 centres which reported using the MIST procedure, premedication practice varied widely, with 58.8% using atropine, 47% using fentanyl, and 35.3% using ketamine[56].
A recent review included two RCTs and four observational studies comparing the use of premedication with no medications in preterm infants ranging in age from 25+6 to 36+6 weeks[57]. Five of these studies had small numbers, while a larger multicentre cohort study[6] enrolled 495 infants but included a variety of medication practices[57]. One RCT compared propofol alone with no premedication, enrolling 78 infants between 28 and 32 weeks gestational age (GA)[58]. This study showed that the premedicated group had significantly lower discomfort scores than the non-premedicated group[58]. The premedicated group experienced more desaturations and required more nasal intermittent mandatory ventilation (nNIMV), but overall, duration of nNIMV was not significantly different between groups[58]. Another small RCT (n=34; 28 to 33+6 weeks GA), comparing fentanyl versus no premedication for LISA showed lower pain scores in infants who received premedication[59]. More desaturations and a higher oxygen requirement were seen in the infants receiving fentanyl, but this effect was not statistically significant[58].
While the data are limited as yet, the use of premedication for all non-urgent laryngoscopies can be recommended based on the principle of ensuring adequate analgesia to avoid the short- and long-term impacts of pain. Fentanyl or remifentanil is recommended for LISA/MIST in addition to a vagolytic such as atropine[15][60]. Due to risk for apnea and chest-wall rigidity, a lower dose may be considered (e.g., fentanyl 1 mcg/kg) and the care team should be prepared to provide positive pressure ventilation, secure an alternate airway, or both.
Surfactant via supraglottic airway
No data were found to support use of premedication for this method of surfactant administration. One review suggested a similar protocol to that used in intubation, based on limited evidence[15].
|
Medication |
Dosing |
Route |
Timing |
Indications |
|
Intubation/INSURE |
||||
|
Atropine |
0.01–0.02 mg/kg |
IV** |
Push |
Prevention of bradycardia |
|
Fentanyl |
2 mcg/kg |
IV** |
Slow push over 2–5 minutes |
Analgesia |
|
Succinylcholine |
2 mg/kg |
IV** |
Push |
Muscle relaxant |
|
LISA/MIST |
||||
|
Atropine |
0.01–0.02 mg/kg |
IV** |
Push |
Prevention of bradycardia |
|
Fentanyl |
1 mcg/kg |
IV** |
Slow push over 2–5 minutes |
Analgesia |
|
*When intravenous (IV) access cannot be obtained, consider intranasal fentanyl at a dose of 1 to 2 mcg/kg. Every attempt should be made to prioritize IV access. **Saline flush should be provided to ensure clearance of line and tubing, especially for drugs of small volume. INSURE Intubate, surfactant, extubate; LISA Less invasive surfactant administration; MIST Minimally invasive surfactant therapy |
||||
Other aspects of endotracheal intubation/laryngoscopy
Best practice requires that all laryngoscopies should be performed or supervised by a skilled and experienced care provider[14]. Decreasing the duration and number of procedural attempts lowers risk for vagal stimulation and airway obstruction, which can lead to apnea and hypoxia[21][27]. Appropriate equipment and skilled personnel should be in place before starting intubation/laryngoscopy. Infants should receive oxygen as required to maintain normal saturations pre-procedure, and practitioners should be ready to provide assisted ventilation when medications take effect. Continuous monitoring of vital signs should be in place, and the duration of each attempt should be limited. Videolaryngoscopy has been shown to improve the success of intubation at first attempt[61][62] and should be the preferred approach[63]. Apneic oxygenation, using high-flow nasal cannula during the intubation procedure with an FiO2 matching the infant’s current requirements, increases the likelihood of successful intubation at first attempt while lowering risk of desaturation[64]. Once placed, tube placement should be confirmed with demonstration of exhaled CO2 and accompanying improvement in heart rate[65]. Newborns with airway anomalies should be intubated by an expert team, with an otolaryngologist aor anesthesiologist in attendance, if possible.
Recommendations
- Premedication should be administered for all laryngoscopies, except in cases of clinical emergency (e.g., neonatal resuscitation). When intravenous (IV) access cannot be obtained, intranasal (IN) fentanyl may be considered.
- Optimal premedication for intubation and surfactant delivery by the INSURE procedure (intubate, surfactant, extubate) should include combining a vagolytic, a fast-acting opiate, and a short-acting muscle relaxant. Suggested drugs and dosages are atropine (0.01 to 0.02 mg/kg), fentanyl 2 mcg/kg (administered slowly over 2 to 5 minutes), and succinylcholine (2 mg/kg). Succinylcholine should not be used in cases of hyperkalemia, family history of malignant hyperthermia, or congenital neuromuscular disorder.
- LISA (less invasive surfactant administration) and MIST (minimally invasive surfactant therapy), should include non-pharmacological measures (e.g., “facilitated tucking” or containment) to manage pain. In addition to non-pharmacological measures, atropine should be administered before MIST and LISA to avoid vagal stimulation and bradycardia, then fentanyl at a lower dose (1 mcg/kg as a slow IV push over 2 to 5 minutes).
- There are insufficient data to make a definite recommendation on the use of premedication for supraglottic airway placement. Similar protocols to those used in intubation may be considered.
- Providers should be aware of the pharmacokinetics of the drugs they use, such that dosing can be adjusted in cases of hepatic or renal dysfunction. Remifentanil is cleared rapidly in blood and tissue and as such is less dependent on hepatic and renal function. Remifentanil may be considered as an alternate to fentanyl in cases of hepatic or renal dysfunction.
Figure 1. Flow diagram to aid premedication decision-making
INSURE Intubate, surfactant, extubate; LISA Less invasive surfactant administration; MIST Minimally invasive surfactant therapy; SGA Supraglottic airway
Acknowledgement
We are grateful to Ms. Charmaine De Castro, Information Specialist at the Sidney Liswood Health Sciences Library at Mount Sinai Hospital, Toronto, for performing the literature search for this review. This statement was reviewed by the Acute Care, Community Paediatrics, and Drug Therapy Committees, and Neonatal-Perinatal Medicine Section Executive, of the Canadian Paediatric Society.
CANADIAN PAEDIATRIC SOCIETY FETUS AND NEWBORN COMMITTEE (2024-2025)
Members: Souvik Mitra MD MSc (Chair), Michael Narvey MD (Past Chair), Sidd Thakore MD (Board Representative), Gabriel Altit MD, Nicole Radziminski MD, Emer Finan MB, Mireille Guillot MD, Frances Morin MD (Resident Member)
Liaisons: William Ehman MD (College of Family Physicians of Canada), Chantal Nelson (Public Health Agency of Canada), Eric Eichenwald MD (American Academy of Pediatrics, Committee on Fetus & Newborn), Douglas Wilson MD (The Society of Obstetricians and Gynaecologists of Canada), Isabelle Milette RN, MScN, NNP (Canadian Association of Neonatal Nurses), Deepak Manhas MD (CPS Neonatal-Perinatal Medicine Section)
Principal authors: Anne-Sophie Gervais MD, Keith J Barrington MD, Emer Finan MB
Funding
There is no funding to declare.
Potential Conflict of Interest
The authors have indicated they have no conflicts of interest.
References
- Caldwell CD, Watterberg KL. Effect of premedication regimen on infant pain and stress response to endotracheal intubation. J Perinatol 2015;35(6):415–8. doi: 10.1038/jp.2014.2272
- Ancora G, Lago P, Garetti E, et al. Evidence‐based clinical guidelines on analgesia and sedation in newborn infants undergoing assisted ventilation and endotracheal intubation. Acta Paediatr 2019;108(2):208–17. doi: 10.1111/apa.14606
- Venkatesh V, Ponnusamy V, Anandaraj J, et al. Endotracheal intubation in a neonatal population remains associated with a high risk of adverse events. Eur J Pediatr 2011;170(2):223–7. doi: 10.1007/s00431-010-1290-8
- Tippmann S, Kidszun A. Adequate analgesia and sedation should be given to neonates during non‐emergency endotracheal intubation. Acta Paediatr 2020;109(1):17–9. doi: 10.1111/apa.14987
- Ng E, Shah V; Canadian Paediatric Society, Fetus and Newborn Committee. Guidelines for surfactant replacement therapy in neonates. Paediatr Child Health 2021;26(1):35-41. doi: 10.1093/pch/pxaa116
- Krajewski P, Szpecht D, Hożejowski R. Premedication practices for less invasive surfactant administration — results from a nationwide cohort study. J Matern Fetal Neonatal Med 2022;35(24):4750–4. doi: 10.1080/14767058.2020.1863365
- Kuek SL, Jacobs SE, Manley BJ. Sedation during minimal invasive surfactant treatment. Acta Paediatr 2020;109(8):1685–6. doi: 10.1111/apa.15228
- Balakrishnan A, Sanghera RS, Boyle EM. New techniques, new challenges—The dilemma of pain management for less invasive surfactant administration? Paediatr Neonatal Pain 2021;3(1):2–8. doi: 10.1002/pne2.12033
- Peterson J, den Boer MC, Roehr CC. To sedate or not to sedate for less invasive surfactant administration: An ethical approach. Neonatol 2021;118(6):639–46. doi: 10.1159/000519283
- Duerden EG, Grunau RE, Guo T, et al. Early procedural pain is associated with regionally-specific alterations in thalamic development in preterm neonates. J Neurosci 2018;38(4):878–86. doi: 10.1523/JNEUROSCI.0867-17.2017
- Duerden EG, Mclean MA, Chau C, et al. Neonatal pain, thalamic development and sensory processing behaviour in children born very preterm. Early Hum Dev 2022;170:105617. doi: 10.1016/j.earlhumdev.2022.105617
- Selvanathan T, Ufkes S, Guo T, et al. Pain exposure and brain connectivity in preterm infants. JAMA Netw Open 2024;7(3):e242551. doi: 10.1001/jamanetworkopen.2024.2551
- McPherson C, Grunau RE. Pharmacologic analgesia and sedation in neonates. Clin Perinatol 2022;49(1):243–65. doi: 10.1016/j.clp.2021.11.014
- Barrington K. Premedication for endotracheal intubation in the newborn infant. Paediatr Child Health. 2011;16(3):159–71. doi: 10.1093/pch/16.3.159
- Durrmeyer X, Walter-Nicolet E, Chollat C, et al. Premedication before laryngoscopy in neonates: Evidence-based statement from the French Society of Neonatology (SFN). Front Pediatr 2023;10:1075184. doi: 10.3389/fped.2022.1075184
- Bottor LT. Rapid sequence intubation in the neonate. Adv Neonatal Care 2009;9(3):111-7; quiz 118-9. doi: 10.1097/ANC.0b013e3181a68cd4
- Lemyre B, Doucette J, Kalyn A, Gray S, Marrin ML. Morphine for elective endotracheal intubation in neonates: A randomized trial [ISRCTN43546373]. BMC Pediatr 2004;4(1):20. doi: 10.1186/1471-2431-4-20
- Perlman JM, Goodman S, Kreusser KL, Volpe JJ. Reduction in intraventricular hemorrhage by elimination of fluctuating cerebral blood-flow velocity in preterm infants with respiratory distress syndrome. New Engl J Med 1985;312(21):1353–7. doi: 10.1056/NEJM198505233122104
- Raju TN, Vidyasagar D, Torres C, Grundy D, Bennett EJ. Intracranial pressure during intubation and anesthesia in infants. J Pediatr 1980;96(5):860–2. doi: 10.1016/s0022-3476(80)80558-0
- Kelly MA, Finer NN. Nasotracheal intubation in the neonate: Physiologic responses and effects of atropine and pancuronium. J Pediatr 1984;105(2):303–9. doi: 10.1016/s0022-3476(84)80137-7
- Maheshwari R, Tracy M, Badawi N, Hinder M. Neonatal endotracheal intubation: How to make it more baby friendly. J Paediatr Child Health 2016;52(5):480–6. doi: 10.1111/jpc.13192
- Marshall TA, Deeder R, Pai S, Berkowitz GP, Austin TL. Physiologic changes associated with endotracheal intubation in preterm infants. Crit Care Med 1984;12(6):501–3. doi: 10.1097/00003246-198406000-00006
- Billingham S. Rapid sequence intubation. J Neonatal Nurs 2012;18(1):25–9. doi.10.1016/j.jnn.2010.11.001
- Badiee Z, Vakiliamini M, Mohammadizadeh M. Remifentanil for endotracheal intubation in premature infants: A randomized controlled trial. J Res Pharm Pract 2013;2(2):75–82. doi: 10.4103/2279-042X.117387
- Ozawa Y, Ades A, Foglia EE, et al. Premedication with neuromuscular blockade and sedation during neonatal intubation is associated with fewer adverse events. J Perinatol 2019;39(6):848–56. doi: 10.1038/s41372-019-0367-0
- Neches SK, Brei BK, Umoren R, et al. Association of full premedication on tracheal intubation outcomes in the neonatal intensive care unit: An observational cohort study. J Perinatol 2023;43(8):1007–14. doi: 10.1038/s41372-023-01632-5
- O’Donnell CPF, Kamlin COF, Davis PG, Morley CJ. Endotracheal intubation attempts during neonatal resuscitation: Success rates, duration, and adverse effects. Pediatrics 2006;117(1):e16-21. doi: 10.1542/peds.2005-0901
- Kumar P, Denson SE, Mancuso TJ, AAP Committee on Fetus and Newborn, Section on Anesthesiology and Pain Medicine. Premedication for nonemergency endotracheal intubation in the neonate. Pediatrics 2010;125(3):608–15. doi: 10.1542/peds.2009-2863
- Barrington KJ, Finer NN, Etches PC. Succinylcholine and atropine for premedication of the newborn infant before nasotracheal intubation: A randomized, controlled trial. Crit Care Med 1989;17(12):1293–6. doi: 10.1097/00003246-198912000-00009
- Afifi J, El-Naggar W, Hatfield T, Sandila N, Baier J, Narvey M. Atropine versus placebo for neonatal nonemergent intubation: A randomized clinical trial. J Pediatr 2025;286:114719. doi: 10.1016/j.jpeds.2025.114719
- de Kort EHM, Hanff LM, Roofthooft D, Reiss IKM, Simons SHP. Insufficient sedation and severe side effects after fast administration of remifentanil during INSURE in preterm newborns. Neonatology 2017;111(2):172–6. doi: 10.1159/000450536
- McNair C, Graydon B, Taddio A. A cohort study of intranasal fentanyl for procedural pain management in neonates. Paediatr Child Health 2018;23(8):e170–5. doi: 10.1093/pch/pxy060
- Ku LC, Simmons C, Smith PB, et al. Intranasal midazolam and fentanyl for procedural sedation and analgesia in infants in the neonatal intensive care unit. J Neonatal Perinatal Med 2019;12(2):143–8. doi: 10.3233/NPM-17149
- Kaushal S, Placencia JL, Maffei SR, Chumpitazi CE. Intranasal fentanyl use in neonates. Hosp Pharm 2020;55(2):126–9. doi: 10.1177/0018578719828335
- Ghanta S, Abdel-Latif ME, Lui K, Ravindranathan H, Awad J, Oei J. Propofol compared with the morphine, atropine, and suxamethonium regimen as induction agents for neonatal endotracheal intubation: A randomized, controlled trial. Pediatrics 2007;119(6):e1248-1255. doi: 10.1542/peds.2006-2708
- Durrmeyer X, Breinig S, Claris O, et al. Effect of atropine with propofol vs atropine with atracurium and sufentanil on oxygen desaturation in neonates requiring nonemergency intubation: A randomized clinical trial. JAMA 2018;319(17):1790–801. doi: 10.1001/jama.2018.3708
- Simons SHP, van der Lee R, Reiss IKM, van Weissenbruch MM. Clinical evaluation of propofol as sedative for endotracheal intubation in neonates. Acta Paediatr 2013;102(11):e487-92. doi: 10.1111/apa.12367
- de Kort EHM, Twisk JWR, van T Verlaat EPG, Reiss IKM, Simons SHP, van Weissenbruch MM. Propofol in neonates causes a dose‐dependent profound and protracted decrease in blood pressure. Acta Paediatr 2020;109(12):2539–46. doi: 10.1111/apa.15282
- Welzing L, Kribs A, Eifinger F, Huenseler C, Oberthuer A, Roth B. Propofol as an induction agent for endotracheal intubation can cause significant arterial hypotension in preterm neonates. Pediatr Anesth 2010;20(7):605–11. doi: 10.1111/j.1460-9592.2010.03330.x
- Avino D, Zhang WH, De Villé A, Johansson AB. Remifentanil versus morphine-midazolam premedication on the quality of endotracheal intubation in neonates: A noninferiority randomized trial. J Pediatr 2014;164(5):1032–7. doi: 10.1016/j.jpeds.2014.01.030
- Ng E, Taddio A, Ohlsson A. Intravenous midazolam infusion for sedation of infants in the neonatal intensive care unit. Cochrane Database Syst Rev 2017;1(1):CD002052. doi: 10.1002/14651858.CD002052.pub3
- Ng E, Klinger G, Shah V, Taddio A. Safety of benzodiazepines in newborns. Ann Pharmacother 2002;36(7–8):1150–5. doi: 10.1345/aph.1A328
- Milési C, Baleine J, Mura T, et al. Nasal midazolam vs ketamine for neonatal intubation in the delivery room: A randomised trial. Arch Dis Child Fetal Neonatal Ed 2018;103(3):F221–6. doi: 10.1136/archdischild-2017-312808
- Wilmott AR, Thompson GC, Lang E, et al. Atropine therapy versus no atropine therapy for the prevention of adverse events in paediatric patients undergoing intubation. Cochrane Database Syst Rev 2018;2018(1):CD010898. doi: 10.1002/14651858.CD010898.pub2
- Pokela ML, Koivisto M. Physiological changes, plasma beta-endorphin and cortisol responses to tracheal intubation in neonates. Acta Paediatr 1994;83(2):151–6. doi: 10.1111/j.1651-2227.1994.tb13040.x
- Truong L, Kim JH, Katheria AC, Finer NN, Marc-Aurele K. Haemodynamic effects of premedication for neonatal intubation: An observational study. Arch Dis Child Fetal Neonatal Ed 2020;105(2):123–7. doi: 10.1136/archdischild-2018-316235
- Thewissen L, Caicedo A, Dereymaeker A, et al. Cerebral autoregulation and activity after propofol for endotracheal intubation in preterm neonates. Pediatr Res 2018;84(5):719–25. doi: 10.1038/s41390-018-0160-3
- Sauer CW, Kong JY, Vaucher YE, et al. Intubation attempts increase the risk for severe intraventricular hemorrhage in preterm infants—A retrospective cohort study. J Pediatr 2016;177:108–13. doi: 10.1016/j.jpeds.2016.06.051
- Cheng C, Tabbara N, Cheng C, Shah V. Intranasal fentanyl for procedural analgesia in preterm infants. Front Pain Res (Lausanne) 2022;2:815014. doi: 10.3389/fpain.2021.815014
- Baleine J, Milési C, Mesnage R, et al. Intubation in the delivery room: Experience with nasal midazolam. Early Hum Dev 2014;90(1):39–43. doi: 10.1016/j.earlhumdev.2013.10.007
- Fahnenstich H, Steffan J, Kau N, Bartmann P. Fentanyl-induced chest wall rigidity and laryngospasm in preterm and term infants. Crit Care Med 2000;28(3):836–9. doi: 10.1097/00003246-200003000-00037
- Müller P, Vogtmann C. Three cases with different presentation of fentanyl-induced muscle rigidity—A rare problem in intensive care of neonates. Am J Perinatol 2000;17(1):23–6. doi: 10.1055/s-2000-7289
- Dewhirst E, Naguib A, Tobias JD. Chest wall rigidity in two infants after low-dose fentanyl administration. Pediatr Emerg Care 2012;28(5):465–8. doi: 10.1097/PEC.0b013e3182535a2a
- Pitsiu M, Wilmer A, Bodenham A, et al. Pharmacokinetics of remifentanil and its major metabolite, remifentanil acid, in ICU patients with renal impairment. Br J Anaesth 2004;92(4):493–503. doi: 10.1093/bja/aeh086
- Elmekkawi A, Abdelgadir D, Van Dyk J, Choudhury J, Dunn M. Use of naloxone to minimize extubation failure after premedication for INSURE procedure in preterm neonates. J Neonatal Perinatal Med 2016;9(4):363–70. doi: 10.3233/NPM-915141
- Brahmbhatt S, Read B, Da Silva O, Bhattacharya S. A survey of minimally invasive surfactant therapy in Canada. Can J Respir Ther 2022;58:122–6. doi: 10.29390/cjrt-2022-011
- Yew R, Fleeman M, Gowda H. Should premedication be used for less invasive surfactant administration (LISA)? Arch Dis Child 2023;108(2):141-3. doi: 10.1136/archdischild-2022-324922
- Dekker J, Lopriore E, van Zanten HA, Tan RNGB, Hooper SB, Te Pas AB. Sedation during minimal invasive surfactant therapy: A randomised controlled trial. Arch Dis Child Fetal Neonatal Ed 2019;104(4):F378–83. doi: 10.1136/archdischild-2018-315015
- Sk H, Saha B, Mukherjee S, Hazra A. Premedication with fentanyl for less invasive surfactant application (LISA): A randomized controlled trial. J Trop Pediatr 2022;68(2):fmac019. doi: 10.1093/tropej/fmac019
- Roberts CT, Halibullah I, Bhatia R, et al. Outcomes after introduction of minimally invasive surfactant therapy in two Australian tertiary neonatal units. J Pediatr 2021;229:141–6. doi: 10.1016/j.jpeds.2020.10.025
- Geraghty LE, Dunne EA, Ní Chathasaigh CM, et al. Video versus direct laryngoscopy for urgent intubation of newborn infants. N Engl J Med 2024;390(20):1885-94. doi: 10.1056/NEJMoa2402785
- Moussa A, Sawyer T, Puia-Dumitrescu M, et al. Does videolaryngoscopy improve tracheal intubation first attempt success in the NICUs? A report from the NEAR4NEOS. J Perinatol 2022;42(9):1210–5. doi: 10.1038/s41372-022-01472-9
- Disma N, Asai T, Cools E, et al. Airway management in neonates and infants: European Society of Anaesthesiology and Intensive Care and British Journal of Anaesthesia joint guidelines. Eur J Anaesthesiol 2024;41(1):3–23. doi: 10.1097/EJA.0000000000001928
- Hodgson KA, Owen LS, Kamlin COF, et al. Nasal high-flow therapy during neonatal endotracheal intubation. N Engl J Med 2022;386(17):1627–37. doi: 10.1056/NEJMoa2116735
- Zaichkin J, Kamath-Rayne BD, Weiner G. The NRP 8th Edition: Innovation in Education. Adv Neonatal Care 2021;21(4):322–32. doi: 10.1097/ANC.0000000000000884
Disclaimer: The recommendations in this position statement do not indicate an exclusive course of treatment or procedure to be followed. Variations, taking into account individual circumstances, may be appropriate. Internet addresses are current at time of publication.