Position statement
Dyslipidemia in children: Diagnosis, evaluation, and management
Posted: Feb 27, 2026
Principal author(s)
Michael Khoury MD, Jean-Luc Bigras MD, Elizabeth A. Cummings MD, Kevin C. Harris MD MHSc, Robert A. Hegele MD, Mélanie Henderson MD PhD, Katherine M. Morrison MD, Julie St-Pierre MD PhD, Peter D. Wong MD PhD, Brian W. McCrindle MD MPH; Canadian Paediatric Society, Community Paediatrics Committee
Abstract
Atherosclerosis begins in youth and is directly linked to the presence and severity of cardiovascular risk factors, including dyslipidemia. Timely identification and management of dyslipidemia in childhood can slow atherosclerotic progression and decrease risk for future cardiovascular disease, especially in children with a genetic disorder predisposing to dyslipidemia (e.g., familial hypercholesterolemia, which is frequently undiagnosed). Existing screening strategies can identify cases of paediatric dyslipidemia effectively but should be conducted both earlier and more broadly. Evaluating for secondary causes of dyslipidemia in children, including medication use and systemic disorders, is essential. First-line therapy centres on lifestyle modifications and dietary changes specific to the dyslipidemia phenotype. Indications for medication depend on the severity of dyslipidemia and assessment for cardiovascular risk on an individual basis.
Keywords: Atherosclerosis; Dyslipidemia; Familial hypercholesterolemia; Hypertriglyceridemia; Lipid disorders
Atherosclerosis, the pathobiological basis of cardiovascular disease (CVD), begins in childhood and is linked directly to both the presence and severity of cardiovascular (CV) risk factors, such as dyslipidemia[1][2]. Early identification and management of dyslipidemia is increasingly imperative, particularly in at-risk populations, such as those predisposed to severe, lifelong dyslipidemias due to inherited lipid disorders such as familial hypercholesterolemia (FH). Yet lipid screening rates to detect such disorders remain low in Canada[3][4]. This Canadian Paediatric Society (CPS) statement encapsulates a longer clinical practice update (CPU) published by the Canadian Cardiovascular Society and Canadian Pediatric Cardiology Association in 2022[5]. It provides an approach to detecting, evaluating, and managing paediatric dyslipidemia with a focus on the early screening and treatment of FH.
Definitions, epidemiology, and genetics
Dyslipidemia is increasingly prevalent among young people in Canada and can result from either inherited, monogenic causes or (increasingly) non-genetic contributors[6]. Normal, borderline, and abnormal paediatric lipoprotein levels are defined in Table 1[7].
|
Table 1. Acceptable, borderline-high, and high fasting plasma lipid and lipoprotein concentrations* |
|||
|
Category |
Acceptable |
Borderline |
Abnormal |
|
Total cholesterol (TC) |
<4.4 mmol/L |
4.4 to <5.2 mmol/L |
≥5.2 mmol/L |
|
Low-density lipoprotein cholesterol (LDL-C) |
<2.8 mmol/L |
2.8 to <3.4 mmol/L |
≥3.4 mmol/L |
|
Non-high-density lipoprotein cholesterol (non-HDL-C) |
<3.10 mmol/L |
3.10 to <3.75 mmol/L |
≥3.75 mmol/L |
|
Triglycerides |
|||
|
0 to 9 years |
<0.8 mmol/L |
0.8 to <1.1 mmol/L |
>1.1 mmol/L |
|
10 to 19 years |
<1.0 mmol/L |
1.0 to <1.5 mmol/L |
>1.5 mmol/L |
|
High-density lipoprotein cholesterol (HDL-C) |
>1.2 mmol/L |
1.0 to 1.2 mmol/L |
<1.0 mmol/L |
|
*While a non-fasting lipid assessment is appropriate for an initial screening assessment, abnormal values should be confirmed with a repeat-fasting lipid assessment. Adapted from reference[7] |
|||
There are 25 monogenic dyslipidemias that present in childhood[8]. FH, the most common of these, is inherited in an autosomal codominant fashion[9]. The heterozygous form has a prevalence of up to 1 in 90 in parts of Quebec and a prevalence of ~1 in 300 in the rest of Canada, occurring in all ancestries[10]. FH is diagnosed either by genetic testing or using phenotypic criteria[10]. A severely elevated low-density lipoprotein cholesterol (LDL-C) cut-point of 4.0 mmol/L has been suggested in children to identify definite FH in the presence of a known causative gene mutation. However, probable FH is considered at the same LDL-C cut-point of 4.0 mmol/L when a first-degree relative with high LDL-C or premature atherosclerotic CVD is present[11].
Other primary paediatric dyslipidemias include severe hypertriglyceridemia[12], combined hyperlipidemia, and elevated lipoprotein(a) (Lp[a]). Severe hypertriglyceridemia can be caused by an autosomal recessive (biallelic) disorder associated with reduced function of either lipoprotein lipase or one of its activating proteins or binding partners[12]. Combined hyperlipidemia, characterized by elevated triglycerides, LDL-C, and apolipoprotein B, is quite common but is most often polygenic[12]. Lp(a) is strongly genetically determined, minimally influenced by secondary factors, and corresponds with increased cardiovascular risk[13][14].
Secondary dyslipidemias represent a growing burden in paediatrics and are summarized in the original CPU (Table 4)[5]. Obesity-related dyslipidemia, which is the most common form of secondary dyslipidemia, typically presents with mild-to-moderate hypertriglyceridemia and low HDL-C, but underlying CV risk relates to increased numbers of small, dense LDL particles[6].
The rationale for early screening and treatment
Atherosclerosis begins in youth, but lipid disorders are often clinically silent throughout childhood and are thus easy to miss. For example, despite the high prevalence of FH[10][15] and the relative ease of diagnosis (i.e., via lipid screening assessment), about 90% of heterozygous FH cases remain unrecognized[9]. Identifying and treating FH early reduces cumulative LDL-C burden (and thus, atherosclerotic burden), with significant, long-term health and socioeconomic benefits[9][16]. Unfortunately, selective screening strategies based on family history still miss between 30% and 60% of dyslipidemic youth[17]. Previous guidelines have recommended universal non-fasting lipid screening for all children between 9 and 11 years of age, with a repeat screen between age 17 and 21, to improve the detection of FH and other heritable lipid disorders[7].
Numerous randomized and prospective observational studies have demonstrated that statin treatment in children with FH effectively lowers their LDL-C and may slow, or even normalize, atherosclerotic progression and CVD significantly[18]-[22]. The evidence-base and rationale for early screening and timely initiation of statin treatment for children with FH are compelling. Of note, the data supporting early treatment of other phenotypes of paediatric dyslipidemia are less robust.
Screening strategies
Using a lipid panel to identify dyslipidemia is simple and easy to include in routine primary care. Two strategies are used in practice: 1) Selective versus universal screening, and 2) Cascade/reverse-cascade screening.
Selective versus universal screening
Selective lipid screening is indicated for children >2 years old who have a positive family history of premature CVD (i.e., angina, myocardial infarction, CAD, or sudden cardiac death in a parent, grandparent, aunt, or uncle at <55 years of age for males and <65 years of age for females)[7]. Selective screening also applies to children with certain medical conditions (e.g., type 2 diabetes mellitus and chronic kidney disease) or risk factors for premature CVD (e.g., obesity, hypertension, smoking)[7][23]. Unfortunately, selective screening is inadequate for the detection of FH. A universal screening strategy, starting somewhat earlier than 9 to 11 years of age[7][24] is recommended as more practical in the Canadian context to detect FH and better prevent lifelong dyslipidemia. Screening should occur after the age of 2 years, however, because LDL-C levels overlap significantly between molecularly confirmed cases of FH and non-cases in children younger than 2 years old[25].
Cascade and reverse-cascade screening
Family-based cascade screening tests first-degree relatives of individuals with FH[26], with the inclusion of genetic testing when available, to identify specific genetic mutations that can influence, variably, the presence and severity of dyslipidemia[9][27]. Integrating FH screening into primary care could not only facilitate FH detection in younger children but in parents (via reverse-cascade screening with or without the inclusion of genetic testing) well before CVD develops[28]. This strategy was shown to be cost-effective in the United Kingdom[29]. Further evaluations in the Canadian context are needed.
Fasting versus non-fasting lipid profile specimens
Measuring non-HDL-C levels without fasting (total cholesterol minus HDL-C) is a simple, appropriate screening test that significantly predicts both the presence and persistence of dyslipidemia because it represents the apo-B-containing lipoproteins[7]. Among the lipoprotein parameters, triglyceride levels vary the most by fasting state. For patients identified to have dyslipidemia on an initial screen, subsequent testing should include fasting evaluations, as outlined below.
Clinical evaluation of paediatric dyslipidemias
Assessing for possible primary and secondary etiologies is part of the clinical evaluation of children and youth with dyslipidemia. Primary genetic dyslipidemias are summarized in Table 3 and selected secondary causes and contributors in Table 4 of the original CPU. Establishing the presence of other risk factors or conditions (Table 2) requires a detailed history and physical examination[6][7][23]. Categorizing risk conditions for premature CVD is instrumental for guiding treatment decisions[30].
Physical examination should include plotting height, weight, and body mass index on standardized growth charts, measuring blood pressure, assessing pubertal stage, excluding goiter or hepatosplenomegaly, and looking for signs of insulin resistance (e.g., acanthosis nigricans, which should prompt a more detailed evaluation for insulin resistance, metabolic syndrome, or both). Physical findings of dyslipidemia (e.g., corneal arcus, xanthelasmas, or tendon xanthomas) all point to homozygous FH and are more rare in children with heterozygous FH or other forms of dyslipidemia[31]. Lipemia retinalis and eruptive xanthomas over extensor surfaces and buttocks suggest significant hypertriglyceridemia[31], while a history of recurrent abdominal pain suggestive of pancreatitis may indicate severe hypertriglyceridemia[32].
Initial biochemical investigation should include a complete blood count, fasting lipid profile, thyroid stimulating hormone (TSH), liver and renal function, urinalysis, fasting glucose, glycated hemoglobin, and other tests guided by clinical assessment. For individuals found to have dyslipidemia, decisions regarding diagnosis and the need for pharmacological therapy should be based on the average of results from at least two fasting lipid profiles obtained at least 2 weeks (but no more than 3 months) apart. From a paediatric perspective, determining Lp(a) levels may further inform cardiovascular risk assessment, though concerns regarding availability of the test, variable assays, and lack of standardization remain[13][33][34]. If a monogenic dyslipidemia (such as FH) is suspected based on the lipid panel pattern (e.g., LDL-C >4.0 mmol/L), consider genetic testing after possible secondary causes of dyslipidemia have been excluded. Genetic testing is typically arranged via paediatric lipid specialists or medical genetics, but is not always available.
Approach to managing dyslipidemia
Diet and lifestyle counselling
Important dietary recommendations for all children and adolescents with dyslipidemia include: 1) maintaining a healthy diet for age according to Canada’s Food Guide, so one high in vegetables, whole fruit, legumes, fish, nuts, vegetable oils, whole grains, milk and yogurt; 2) more commonly choosing plant-based protein sources (e.g., tofu); 3) avoiding trans-fats; 4) lowering intake of saturated fats; and 5) limiting intake of highly processed foods. Consulting with a registered dietitian is strongly recommended, although access to such specialists is often difficult. Encouraging adherence to Canada’s Movement guidelines for children and youth[35] and supporting smoking cessation are also key[36].
Reducing LDL-C
The dietary approach to addressing elevated LDL-C is graded[7][37]. For example, alongside adhering to Canada’s Food Guide recommendations and incorporating dietary patterns from the Mediterranean Diet, total calories from fat should be reduced to less than 30% (but not less than 20%) and holding saturated fats at 8% to 10% of daily caloric intake[7]. Should elevations in LDL-C persist, further reducing saturated fats to <7% and monounsaturated fat to <10% of total daily calories is recommended.
Phytosterols (sterols and stanols) are bioactive compounds found in vegetables such as broccoli, red onion, and carrot. Ingesting 2 g/day of phytosterols (which typically requires supplementation) has been shown to reduce LDL-C between 8% and 10%[38], even in individuals on statin therapy[39]. For these same individuals, supplementing with psyllium, a soluble fibre, can reduce LDL-C in a dose-dependent manner between 5% and 10%[40]. For children with hypercholesterolemia, incorporating plant sterols (2 g daily) and psyllium fibre (6 g/day; 2 to 12 years and 12 g/day >12 years) can be considered[7].While dietary phytosterols and fibre supplementation are effective in lowering LDL-C, their impact on CVD risk is not known.
Triglycerides
Children and adolescents with moderately elevated triglycerides typically respond to lifestyle changes, including a diet low in sugar and refined carbohydrates (in favour of complex carbohydrates), low in fat, and high in fiber[7][41]. Increasing intake of foods rich in omega-3 fatty acids should also be considered[42]. Regular physical activity is recommended, and secondary causes of elevated triglyceride levels such as obesity or uncontrolled diabetes must be addressed. Encourage engagement in multi-disciplinary obesity management programs when needed. Restricting fat significantly is important for children and adolescents with lipoprotein lipase deficiency and severe hypertriglyceridemia (>10 mmol/L) to prevent pancreatitis[41].
Pharmacological therapy
Consideration of drug therapy for dyslipidemia should follow a reasonable trial of dietary and lifestyle modifications (typically 6 months, but this step can be abbreviated for severe dyslipidemia [i.e., LDL-C ≥4.9 mmol/L]). In paediatrics, medication use is reserved for individuals with severe, persistent dyslipidemia and other risk conditions or factors, most commonly FH (Table 2). When indicated, pharmacological therapy should be utilized concomitantly with lifestyle management.
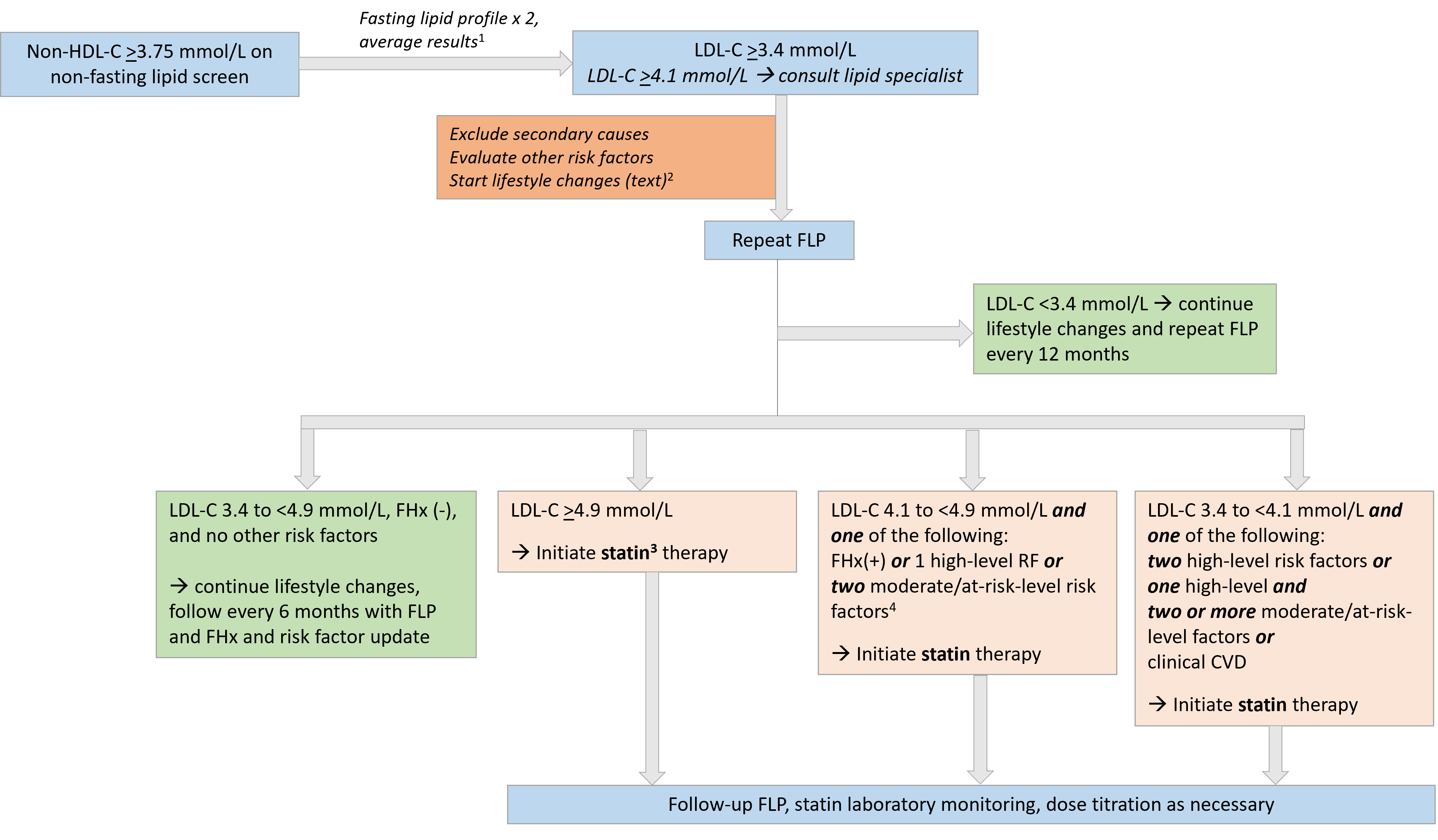
For LDL-C management (most commonly in the setting of FH), statins are the first-line medication and are often started when children are between 8 and 12 years old[5][43]. Recommendations are provided in Figure 1, including thresholds to initiate treatment based on underlying risk. Notably, individuals with type 1 or 2 diabetes have lower LDL-C thresholds for starting statin therapy. Periodic monitoring of liver enzymes for safety, along with muscle-related and other symptoms, counselling on pregnancy prevention and drug interactions, and reinforcing lifestyle measures are all recommended in Figure 3 of the original CPU. Shared decision-making with the family about when best to start medication is required, which involves education, anticipatory counselling, and addressing specific concerns. Pharmacological management of children with homozygous FH should begin immediately upon diagnosis and be led by a lipid specialist (usually a paediatric cardiologist or endocrinologist).
Figure 1. Paediatric statin thresholds for treatment
1. Repeat FLP between 2 weeks and 3 months of initial testing
2. Consider use of dietary supplements (phytosterols and psyllium)
3. Statins typically are initiated starting at 8-12 years of age. Note that statin treatment thresholds in the paediatric population are largely based on expert opinion
4. See Table 2 for high-, moderate, and at-risk level risk factors and conditions.
BMI Body mass index; CVD Cardiovascular disease; FHx (-) or (+) Family history negative or positive; FLP Fasting lipid profile; LDL-C Low-density lipoprotein cholesterol; Non-HDL-C Non-high-density lipoprotein cholesterol
Source: This figure was published in the Canadian Journal of Cardiology 38(8), Khoury M, Bigras JL, Cummings EA, et al. The detection, evaluation, and management of dyslipidemia in children and adolescents: A Canadian Cardiovascular Society/Canadian Pediatric Cardiology Association clinical practice update:1168-79. © Elsevier 2022. It is reproduced with permission.
Statins have similar short- to medium-term safety profiles when used for children aged 8 years and older as for adults[18], and safety data published in a 20-year follow-up study in 2019 were reassuring[22]. The most recent Cochrane review evaluating statin use in children with FH demonstrated little or no difference between treatment and placebo related to liver function, creatinine kinase, myopathy, sex hormone levels or puberty status, or clinical adverse events[18]. No clinical trials to date in children and adolescents have reported rhabdomyolysis (degeneration of skeletal muscle tissue) caused by statin treatment[7]. Nonetheless, both the use of statins in children younger than 8 years old and the long-term safety of statin use in all children require ongoing study. Risks for developing insulin resistance or type 2 diabetes mellitus due to statin use have gained increased attention in recent years. In adults, meta-analysis data have estimated that treating 255 adults with statins for 4 years would result in one case of type 2 diabetes mellitus[44]. While the risk of new-onset diabetes mellitus in paediatric patients treated with statins requires further study, the same 20-year follow-up study noted above found that 1 in 184 individuals with heterozygous FH and treated with statins developed type 2 diabetes mellitus, compared with 2/77 unaffected siblings[22], providing early reassuring data regarding the risk of diabetes mellitus development for children receiving statin treatment. Of note, emerging clinical trial data may soon provide evidence supporting use of novel lipid-lowering drugs, including biologic agents that target proprotein convertase subtilisin/kexin type 9 (PCSK9)[45][46] and targets that act independent of the LDL receptor in the paediatric age group[47], thereby broadening treatment options for patients with severely elevated LDL-C from genetic dyslipidemias.
For individuals with hypertriglyceridemia, dietary supplementation with omega-3 fatty acids may have benefit. In adults, pharmacological doses of 2 to 4 g per day of long-chain omega-3 fatty acids reduced triglyceride levels by up to 30% [41], and the use of icosapent ethyl (the ethyl ester of eicosapentaenoic acid [EPA]) lowered CVD events (also in adults and compared with mineral oil) in the REDUCE-IT trial[48]. However, other omega-3 fatty acid prescription formulations that contain both EPA and docosahexaenoic acid (DHA) did not reduce CVD events to a similar extent[49]. Also, small, randomized trials of omega-3 fatty acid treatment in children and adolescents have not shown statistically significant improvements in triglyceride levels compared with placebo[50].
Prescription omega-3 fatty acid formulations should be used in children and adolescents because over-the-counter supplements often have lower levels of EPA and DHA, may not have undergone comparable regulatory safety monitoring, and may contain potentially harmful contaminants such as toxins and oxidized fatty acids[51]. Individuals with fish allergies or specific dietary preferences should consult their physician regarding suitable omega-3 formulations. Further discussion regarding the pharmacological treatment of hypertriglyceridemia is included in the original CPU[5]. Generally, treating persistent hypertriglyceridemia (plasma triglyceride 2.3 to 5.5 mmol/L despite lifestyle interventions) or severe hypertriglyceridemia (>5.5 mmol/L) with medication, including the use of prescription omega-3 fatty acids or fibrates, can be considered alongside strict dietary management, although evidence of benefit and safety are limited in children. Evaluation and management by a lipid specialist is recommended, particularly when fibrate medications are considered, due to their substantial drug interactions and adverse effect profile.
Conclusion
Paediatric lipid disorders, including genetic disorders such as FH that result in marked dyslipidemia, are common and increase the risk for premature cardiovascular disease. Treatment of FH from childhood can normalize future cardiovascular risk. These disorders are typically clinically silent and require a lipid screening assessment for diagnosis. An approach that incorporates universal, cascade, and selective screening is the most comprehensive strategy to detect at-risk children and permit timely treatment.
Key points and recommendations
- Atherosclerosis begins in youth. Its presence and severity are linked to cardiovascular risk factors, including dyslipidemia.
- Familial hypercholesterolemia (FH) is common (~1:300) and easy to screen for using low-density lipoprotein cholesterol (LDL-C) testing, but remains vastly underdiagnosed in Canada.
- Universal lipid screening between 2 and 10 years of age is recommended, using a fasting or non-fasting non-high-density lipoprotein cholesterol (non-HDL-C) or an LDL-C test. In cases of definite or probable FH, this screen should be coupled with cascade screening of family members to identify other affected individuals. Selective screening at any time should be considered for children with identified cardiovascular (CV) risk factors, medical conditions predisposing to increased risk, or a positive family history of premature cardiovascular disease (CVD) or dyslipidemia.
- A thorough history and physical examination, with additional investigations as needed, are required to exclude secondary causes of paediatric dyslipidemia.
- Lifestyle and dietary management are first-line treatment strategies for nearly all cases of paediatric dyslipidemia. Counselling includes Canada’s Food Guide and the Canadian Society for Exercise Physiology’s Movement Guidelines for Children and Youth recommendations.
- When lipid-lowering medications are started, lifestyle and dietary management continue to be important.
- Clinical diagnosis and shared decision-making regarding the need for pharmacological therapy are based on the average of results from at least two fasting lipid profiles obtained at least 2 weeks but no more than 3 months apart.
- Referral to a paediatric lipid specialist can facilitate lifestyle or pharmacotherapy management, especially when there is marked dyslipidemia at diagnosis (LDL-C >4.1 mmol/L or triglyceride levels >5.5 mmol/L) or dyslipidemia in the setting of risk factors or conditions (Table 2).
- Statin therapy can be considered between ages 8 and 12 years when a child’s LDL-C remains above threshold despite lifestyle management (Figure 1). Routine safety monitoring and LDL-C treatment targets are essential to care and management.
Acknowledgements
This position statement was reviewed by the Adolescent Health and Nutrition and Gastroenterology Committees of the Canadian Paediatric Society.
CANADIAN PAEDIATRIC SOCIETY COMMUNITY PAEDIATRICS COMMITTEE (2023-2024)
Members: Peter Wong MD (Chair), Jill Borland Starkes MD (Board Representative), Michael Hill MD, Audrey Lafontaine MD, Meta van den Heuvel MD, Kelcie Lahey MD MSc
Liaisons: Richa Agnihotri MD (CPS Community Paediatrics Section)
Principal authors: Michael Khoury MD, Jean-Luc Bigras MD, Elizabeth A. Cummings MD, Kevin C. Harris MD MHSc, Robert A. Hegele MD, Mélanie Henderson MD PhD, Katherine M. Morrison MD, Julie St-Pierre MD PhD, Peter D. Wong MD PhD, Brian W. McCrindle MD MPH
Funding
There is no funding to declare.
Potential Conflict of Interest
MK has served on advisory boards for Ultragenyx.
BWM is a consultant for Amryt Pharma, Chiesi, Esperion and Ultragenyx.
RH reported receiving honoraria from Amgen, HLS Therapeutics and Sanofi. Is a member Amgen, HLS Therapeutics, Novartis, Pfizer, Sanofi, Acasti, Aegerion, Akoea and Arrowhead Scientific Advisory Boards. Has received funding or grants from Amgen, Arrowhead, The Medicines Company, LIB Therapeutics, Novartis and Regeneron.
MH reported receiving research grants from CIHR, Diabète Québec, FRQS and CMDO. Is an unpaid member of the Comité scientifique sur la prévention de l'obésité and Institut national de santé publique du Québec.
KM is a member of the Novo Nordisk and Rhythm Pharmaceuticals advisory Boards. Is a member of the Novartis Data Safety Monitoring Board.
JSP reported receiving Obesity Educational Grants from Novo Nordisk and Lilly. Is a member of the Novo Nordisk Global Best Clinical Management Committee.
No other disclosures were reported.
References
- Berenson GS, Srinivasan SR, Bao W, Newman WP, Tracy RE, Wattigney WA. Association between multiple cardiovascular risk factors and atherosclerosis in children and young adults. The Bogalusa Heart Study. N Engl J Med 1998;338(23):1650-6. doi: 10.1056/NEJM199806043382302
- McGill HC, McMahan CA, Zieske AW, et al. Associations of coronary heart disease risk factors with the intermediate lesion of atherosclerosis in youth. The Pathobiological Determinants of Atherosclerosis in Youth (PDAY) Research Group. Arterioscler Thromb Vasc Biol 2000;20(8):1998-2004. doi: 10.1161/01.atv.20.8.1998
- Khoury M, Rodday AM, Mackie AS, et al. Pediatric lipid screening and treatment in Canada: Practices, attitudes, and barriers. Can J Cardiol 2020;36(9):1545-9. doi: 10.1016/j.cjca.2020.05.035
- Christian S, Ridsdale R, Lin M, Khoury M. Evaluating the prevalence of lipid assessments in children in Alberta, Canada. CMAJ Open 2023;11(5):E820-E825. doi: 10.9778/cmajo.20220163
- Khoury M, Bigras JL, Cummings EA, et al. The detection, evaluation, and management of dyslipidemia in children and adolescents: A Canadian Cardiovascular Society/Canadian Pediatric Cardiology Association clinical practice update. Can J Cardiol 2022;38(8):1168-79. doi: 10.1016/j.cjca.2022.05.002
- Lazarte J, Hegele RA. Pediatric dyslipidemia—Beyond familial hypercholesterolemia. Can J Cardiol 2020;36(9):1362-71. doi: 10.1016/j.cjca.2020.03.020
- Expert Panel on Integrated Guidelines for Cardiovascular Health and Risk Reduction in Children and Adolescents; National Heart, Lung, and Blood Institute. Summary report. Pediatrics 2011;128(Suppl 5):S213-56. doi: 10.1542/peds.2009-2107C
- Hegele RA, Borén J, Ginsberg HN, et al. Rare dyslipidaemias, from phenotype to genotype to management: A European Atherosclerosis Society task force consensus statement. Lancet Diabetes Endocrinol 2020;8(1):50-67. doi: 10.1016/S2213-8587(19)30264-5
- Wiegman A, Gidding SS, Watts GF, et al. Familial hypercholesterolaemia in children and adolescents: Gaining decades of life by optimizing detection and treatment. Eur Heart J 2015;36(36):2425-37. doi: 10.1093/eurheartj/ehv157
- Brunham LR, Ruel I, Aljenedil S, et al. Canadian Cardiovascular Society position statement on familial hypercholesterolemia: Update 2018. Can J Cardiol 2018;34(12):1553-63. doi: 10.1016/j.cjca.2018.09.005
- Ruel I, Brisson D, Aljenedil S, et al. Simplified Canadian definition for familial hypercholesterolemia. Can J Cardiol 2018;34(9):1210-14. doi: 10.1016/j.cjca.2018.05.015
- Dron JS, Hegele RA. Genetics of hypertriglyceridemia. Front Endocrinol (Lausanne) 2020;11:455. doi: 10.3389/fendo.2020.00455
- Tsimikas S. A test in context: Lipoprotein(a); Diagnosis, prognosis, controversies, and emerging therapies. J Am Coll Cardiol 2017;69(6):692-711. doi: 10.1016/j.jacc.2016.11.042
- Raitakari O, Kartiosuo N, Pahkala K, et al. Lipoprotein(a) in youth and prediction of major cardiovascular outcomes in adulthood. Circulation 2023;147(1):23-31. doi: 10.1161/circulationaha.122.060667
- de Ferranti SD, Rodday AM, Mendelson MM, Wong JB, Leslie LK, Sheldrick RC. Prevalence of familial hypercholesterolemia in the 1999 to 2012 United States National Health and Nutrition Examination Surveys (NHANES). Circulation 2016;133(11):1067-72. doi: 10.1161/circulationaha.115.018791
- Defesche JC, Gidding SS, Harada-Shiba M, Hegele RA, Santos RD, Wierzbicki AS. Familial hypercholesterolaemia. Nat Rev Dis Primers 2017;3:17093. doi: 10.1038/nrdp.2017.93
- Haney EM, Huffman LH, Bougatsos C, Freeman M, Steiner RD, Nelson HD. Screening and treatment for lipid disorders in children and adolescents: Systematic evidence review for the US Preventive Services Task Force. Pediatrics 2007;120(1):e189-214. doi: 10.1542/peds.2006-1801
- Vuorio A, Kuoppala J, Kovanen PT, et al. Statins for children with familial hypercholesterolemia. Cochrane Database Syst Rev 2019;2019(11):CD006401. doi: 10.1002/14651858.CD006401.pub5
- Khoury M, McCrindle BW. The rationale, indications, safety, and use of statins in the pediatric population. Can J Cardiol 2020;36(9):1372-83. doi: 10.1016/j.cjca.2020.03.041
- de Jongh S, Lilien MR, op't Roodt J, Stroes ESG, Bakker HD, Kastelein JJP. Early statin therapy restores endothelial function in children with familial hypercholesterolemia. J Am Coll Cardiol 2002;40(12):2117-21. doi: 10.1016/s0735-1097(02)02593-7
- Wiegman A, Hutten BA, de Groot E, et al. Efficacy and safety of statin therapy in children with familial hypercholesterolemia: A randomized controlled trial. JAMA 2004;292(3):331-7. doi: 10.1001/jama.292.3.331
- Luirink IK, Wiegman A, Kusters DM, et al. 20-year follow-up of statins in children with familial hypercholesterolemia. N Engl J Med 2019;381(16):1547-56. doi: 10.1056/NEJMoa1816454
- Khoury M, Kavey REW, St Pierre J, McCrindle BW. Incorporating risk stratification into the practice of pediatric preventive cardiology. Can J Cardiol 2020;36(9):1417-28. doi: 10.1016/j.cjca.2020.06.025
- Goldberg AC, Hopkins PN, Toth PP, et al. Familial hypercholesterolemia: Screening, diagnosis and management of pediatric and adult patients; Clinical guidance from the National Lipid Association Expert Panel on Familial Hypercholesterolemia. J Clin Lipidol 2011;5(3):133-40. doi: 10.1016/j.jacl.2011.03.001
- Vuorio AF, Turtola H, Kontula K. Neonatal diagnosis of familial hypercholesterolemia in newborns born to a parent with a molecularly defined heterozygous familial hypercholesterolemia. Arterioscler Thromb Vasc Biol 1997;17(11):3332-7. doi: 10.1161/01.atv.17.11.3332
- Wald DS, Bestwick JP. Reaching detection targets in familial hypercholesterolaemia: Comparison of identification strategies. Atherosclerosis 2020;293:57-61. doi: 10.1016/j.atherosclerosis.2019.11.028
- Brown EE, Sturm AC, Cuchel M, et al. Genetic testing in dyslipidemia: A scientific statement from the National Lipid Association. J Clin Lipidol 2020;14(4):398-413. doi: 10.1016/j.jacl.2020.04.011
- Wald DS, Bestwick JP, Morris JK, Whyte K, Jenkins L, Wald NJ. Child-parent familial hypercholesterolemia screening in primary care. N Engl J Med 2016;375(17):1628-37. doi: 10.1056/NEJMoa1602777
- McKay AJ, Hogan H, Humphries SE, Marks D, Ray KK, Miners A. Universal screening at age 1-2 years as an adjunct to cascade testing for familial hypercholesterolaemia in the UK: A cost-utility analysis. Atherosclerosis 2018;275:434-43. doi: 10.1016/j.atherosclerosis.2018.05.047
- de Ferranti SD, Steinberger J, Ameduri R, et al. Cardiovascular risk reduction in high-risk pediatric patients: A scientific statement from the American Heart Association. Circulation 2019;139(13):e603-e634. doi: 10.1161/CIR.0000000000000618
- Descamps OS, Tenoutasse S, Stephenne X, et al. Management of familial hypercholesterolemia in children and young adults: Consensus paper developed by a panel of lipidologists, cardiologists, paediatricians, nutritionists, gastroenterologists, general practitioners and a patient organization. Atherosclerosis 2011;218(2):272-80. doi: 10.1016/j.atherosclerosis.2011.06.016
- Shah AS, Wilson DP. Primary hypertriglyceridemia in children and adolescents. J Clin Lipidol 2015;9(5 Suppl):S20-8. doi: 10.1016/j.jacl.2015.04.004
- Alonso R, Andres E, Mata N, et al. Lipoprotein(a) levels in familial hypercholesterolemia: An important predictor of cardiovascular disease independent of the type of LDL receptor mutation. J Am Coll Cardiol 2014;63(19):1982-9. doi: 10.1016/j.jacc.2014.01.063
- Ellis KL, Pérez de Isla L, Alonso R, Fuentes F, Watts GF, Mata P. Value of measuring lipoprotein(a) during cascade testing for familial hypercholesterolemia. J Am Coll Cardiol 2019;73(9):1029-39. doi: 10.1016/j.jacc.2018.12.037
- Canadian Society for Exercise Physiology. Canadian 24-Hour Movement Guidelines for Children and Youth: An Integration of Physical Activity, Sedentary Behaviour, and Sleep. (Accessed October 3, 2025).
- Harvey J, Chadi N. Strategies to promote smoking cessation among adolescents. Paediatr Child Health 2016;21(4):201-8. doi: 10.1093/pch/21.4.201
- Grundy SM, Stone NJ, Bailey AL, et al. 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA Guideline on the management of blood cholesterol: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation 2019;139(25):e1082-e1143. doi: 10.1161/CIR.0000000000000625
- Katan MB, Grundy SM, Jones P, et al; Stresa Workshop Participants. Efficacy and safety of plant stanols and sterols in the management of blood cholesterol levels. Mayo Clin Proc 2003;78(8):965-78. doi: 10.4065/78.8.965
- Han S, Jiao J, Xu J, et al. Effects of plant stanol or sterol-enriched diets on lipid profiles in patients treated with statins: Systematic review and meta-analysis. Sci Rep 2016;6:31337. doi: 10.1038/srep31337
- Brum J, Ramsey D, McRorie J, Bauer B, Kopecky SL. Meta-analysis of usefulness of psyllium fiber as adjuvant antilipid therapy to enhance cholesterol lowering efficacy of statins. Am J Cardiol 2018;122(7):1169-74. doi: 10.1016/j.amjcard.2018.06.040
- Mach F, Baigent C, Catapano AL, et al. 2019 ESC/EAS guidelines for the management of dyslipidaemias: Lipid modification to reduce cardiovascular risk. Eur Heart J 2020;41(1):111-88. doi: 10.1093/eurheartj/ehz455
- Abdelhamid AS, Brown TJ, Brainard JS, et al. Omega-3 fatty acids for the primary and secondary prevention of cardiovascular disease. Cochrane Database Syst Rev 2020;3(3):CD003177. doi: 10.1002/14651858.CD003177.pub5
- Rodenburg J, Vissers MN, Wiegman A, et al. Statin treatment in children with familial hypercholesterolemia: The younger, the better. Circulation 2007;116(6):664-8. doi: 10.1161/CirculationAHA.106.671016
- Sattar N, Preiss D, Murray HM, et al. Statins and risk of incident diabetes: A collaborative meta-analysis of randomised statin trials. Lancet 2010;375(9716):735-42. doi: 10.1016/S0140-6736(09)61965-6
- Santos RD, Ruzza A, Hovingh GK, et al. Evolocumab in pediatric heterozygous familial hypercholesterolemia. N Engl J Med 2020;383(14):1317-27. doi: 10.1056/NEJMoa2019910
- Daniels S, Caprio S, Chaudhari U, et al. PCSK9 inhibition with alirocumab in pediatric patients with heterozygous familial hypercholesterolemia: The ODYSSEY KIDS study. J Clin Lipidol 2020;14(3):322-30.e5. doi: 10.1016/j.jacl.2020.03.001
- Wiegman A, Greber-Platzer S, Ali S, et al. Evinacumab for pediatric patients with homozygous familial hypercholesterolemia. Circulation 2024;149(5):343-53. doi: 10.1161/CirculationAHA.123.065529
- Bhatt DL, Steg PG, Miller M, et al. Cardiovascular risk reduction with icosapent ethyl for hypertriglyceridemia. N Engl J Med 2019;380(1):11-22. doi: 10.1056/NEJMoa1812792
- Nicholls SJ, Lincoff AM, Garcia M, et al. Effect of high-dose omega-3 fatty acids vs corn oil on major adverse cardiovascular events in patients at high cardiovascular risk: The STRENGTH randomized clinical trial. JAMA 2020;324(22):2268-80. doi: 10.1001/jama.2020.22258
- de Ferranti SD, Milliren CE, Denhoff ER, et al. Using high-dose omega-3 fatty acid supplements to lower triglyceride levels in 10- to 19-year-olds. Clin Pediatr (Phila) 2014;53(5):428-38. doi: 10.1177/0009922814528032
- Fialkow J. Omega-3 fatty acid formulations in cardiovascular disease: Dietary supplements are not substitutes for prescription products. Am J Cardiovasc Drugs 2016;16(4):229-39. doi: 10.1007/s40256-016-0170-7
Disclaimer: The recommendations in this position statement do not indicate an exclusive course of treatment or procedure to be followed. Variations, taking into account individual circumstances, may be appropriate. Internet addresses are current at time of publication.