Position statement

Guidelines for detection, management and prevention of hyperbilirubinemia in term and late preterm newborn infants
Posted: Jun 1, 2007 | Reaffirmed: Feb 28, 2018
Principal author(s)
KJ Barrington, K Sankaran; Canadian Paediatric Society, Fetus and Newborn Committee
Abridged version: Paediatr Child Health 2007;12(Suppl B):1B-12B
Abstract
Hyperbilirubinemia is very common and usually benign in the term newborn infant and the late preterm infant at 35 to 36 completed weeks’ gestation. Critical hyperbilirubinemia is uncommon but has the potential for causing long-term neurological impairment. Early discharge of the healthy newborn infant, particularly those in whom breastfeeding may not be fully established, may be associated with delayed diagnosis of significant hyperbilirubinemia. Guidelines for the prediction, prevention, identification, monitoring and treatment of severe hyperbilirubinemia are presented.
Key Words: 35 weeks’ gestation; Hyperbilirubinemia; Jaundice; Preterm newborn; Term newborn
Background and epidemiology
Definitions of terms as used in this statement
- Kernicterus – the pathological finding of deep-yellow staining of neurons and neuronal necrosis of the basal ganglia and brainstem nuclei.
- Acute bilirubin encephalopathy – a clinical syndrome, in the presence of severe hyperbilirubinemia, of lethargy, hypotonia and poor suck, which may progress to hypertonia (with opisthotonos and retrocollis) with a high-pitched cry and fever, and eventually to seizures and coma.
- Chronic bilirubin encephalopathy – the clinical sequelae of acute encephalopathy with athetoid cerebral palsy with or without seizures, developmental delay, hearing deficit, oculomotor disturbances, dental dysplasia and mental deficiency [1]-[3].
- Severe hyperbilirubinemia – a total serum bilirubin (TSB) concentration greater than 340 µmol/L at any time during the first 28 days of life.
- Critical hyperbilirubinemia – a TSB concentration greater than 425 µmol/L during the first 28 days of life.
The prevention, detection and management of jaundice in otherwise healthy term and late preterm newborn infants remain a challenge, partly because jaundice is so common and kernicterus is so rare in comparison [4]-[6]. It is estimated that 60% of term newborns develop jaundice and 2% reach a TSB concentration greater than 340 µmol/L [7]. Acute encephalopathy does not occur in full-term infants whose peak TSB concentration remains below 340 µmol/L and is very rare unless the peak TSB concentration exceeds 425 µmol/L. Above this level, the risk for toxicity progressively increases [8][9]. More than three-quarters of the infants in the United States’ kernicterus registry (between 1992 and 2002) had a TSB concentration of 515 µmol/L or greater, and two-thirds had a concentration exceeding 600 µmol/L [10]. Even with concentrations greater than 500 µmol/L, there are still some infants who will escape encephalopathy. All of the reasons for the variable susceptibility of infants are not known; however, dehydration, hyperosmolarity, respiratory distress, hydrops, prematurity, acidosis, hypoalbuminemia, hypoxia and seizures are said to increase the risk of acute encephalopathy in the presence of severe hyperbilirubinemia [9][11], although reliable evidence to confirm these associations is lacking [12]. In addition, some infants with severe hyperbilirubinemia are found to have sepsis, but both sepsis and hyperbilirubinemia are common in the neonatal period, and sepsis appears to be uncommon in the well-appearing infant with severe hyperbilirubinemia.
Milder degrees of hyperbilirubinemia not leading to a clinical presentation of acute encephalopathy may also be neurotoxic and cause less severe long-term complications. This remains controversial; however, if there are bilirubin concentrations at which subtle cerebral injury can occur, the thresholds are unknown [13]-[15]. The collaborative perinatal project, examining 54,795 live births in the United States, was unable to find any consistent association between peak TSB concentrations below critical levels and IQ or other adverse outcomes [12]. Therefore, prevention of acute encephalopathy remains the justification for the prevention, detection and treatment of severe hyperbilirubinemia [16][17].
The incidence of acute encephalopathy is uncertain, but it continues to occur. The Canadian Paediatric Surveillance Program (CPSP) recently reported 258 full-term infants over a two-year period (2002 to 2004) who either required exchange transfusion or had critical hyperbilirubinemia (excluding infants with rhesus isoimmunization) [18]. Twenty per cent of these infants had at least one abnormal neurological sign at presentation, and 5% had documented hearing loss or significant neurological sequelae at discharge. During this period, the live birth rate in Canada was approximately 330,000 per year, leading to a calculated minimal incidence of this degree of severity of hyperbilirubinemia of approximately four in 10,000 live births. If we assume that the entire 20% of infants with neurological findings at presentation had acute bilirubin encephalopathy, the incidence of this complication would be one in 10,000 live births, an incidence similar to that of phenylketonuria. The incidence of chronic encephalopathy is also uncertain, but it has been estimated to be approximately one in 100,000 [19][20]. This situation occurs despite the fact that a large number of infants already receive intensive preventive therapy [11]. The CPSP report [18] noted that 13 of the infants continued to have important neurological abnormalities at final discharge, suggesting a chronic bilirubin encephalopathy incidence of one in 50,000, similar to the frequency reported from a Danish study [21].
Acute bilirubin encephalopathy was first recognized in infants with rhesus hemolytic disease; this etiology is now largely avoidable and, consequently, has become rare. Reports [22][23] indicate that acute bilirubin encephalopathy continues to occur in otherwise healthy infants with, and occasionally without, identifiable risk factors. Prevention of this rare but serious disease requires appropriate clinical assessment, interpretation of TSB concentration and treatment, which must include all systems involved in the provision of health care and community support.
Several risk factors have been identified for the development of severe hyperbilirubinemia in the newborn (Table 1). These risk factors are all common and the attributable risk of each is therefore very low. They are of limited use in directing surveillance, investigation or therapy by themselves, but can be useful in combination with timed TSB analysis. It should also be noted that although a large number of studies have demonstrated an increased risk of severe hyperbilirubinemia with breastfeeding, one study [24] found that exclusive breastfeeding was associated with a lower incidence of hyperbilirubinemia. This may represent cultural differences in the approach to breastfeeding and the support mechanisms in place.
| TABLE 1 Risk factors for the development of severe hyperbilirubinemia |
|
|
Risk Factor |
Approximate odds ratio in comparison with the rest of the population |
|
Visible jaundice at younger than 24 h |
Unclear |
|
Visible jaundice before discharge at any age |
Unclear |
|
Shorter gestation (less than 38 weeks) |
For 36 weeks, 1.9 to 7.7 |
|
Previous sibling with severe hyperbilirubinemia |
4.8 |
|
Visible bruising |
2.6 |
|
Cephalhematoma |
3.6 |
|
Male sex |
1.3 to 1.7 |
|
Maternal age older than 25 years of age |
2.6 |
|
Asian or European background |
5.2 or 1.2, respectively |
|
Dehydration |
Depends on severity |
|
Exclusive and partial breastfeeding |
Very variable in the literature |
Purpose of the statement
The aim of the present statement was to develop guidelines for practice based on evidence-based answers to the following questions:
- Can severe hyperbilirubinemia be accurately predicted?
- Who should have their bilirubin concentration measured, when and by what method?
- How can the risk of severe hyperbilirubinemia be reduced?
- When should severe hyperbilirubinemia be treated?
Methods of statement development
A search was carried out in MEDLINE and the Cochrane library and was last updated in January 2007. Search terms in MEDLINE were hyperbilirubinemia and newborn, and the clinical queries filter of Haynes et al [25] was applied using the broad, sensitive option. Other searches without the filter were carried out to find publications addressing specific issues. The hierarchy of evidence from the Centre for Evidence-Based Medicine was applied using levels of evidence for both treatment and prognosis [26] (Table 2). The reference lists of recent publications were also examined – in particular, the evidence-based review by Ip et al [16] and a more extensive review by the same author [12] performed for the Agency for Healthcare Research and Quality of the US Department of Health and Human Services. The references of the recent statement of the American Academy of Pediatrics [11] were also examined.
| TABLE 2 Levels of Evidence used in this statement |
|||
|
Level |
Therapy |
Prognosis |
Diagnosis |
|
1a |
SR (with homogeneity) of RCTs |
SR (with homogeneity) of inception cohort studies |
SR (with homogeneity) of level 1 diagnostic studies |
|
1b |
Individual RCT (with narrow CI) |
Individual inception cohort study with 80% or greater follow-up |
Validating cohort study with good reference standards |
|
2a |
SR (with homogeneity) of cohort studies |
SR (with homogeneity) of either retrospective cohort studies or untreated control groups in RCTs |
SR (with homogeneity) of level greater than 2 diagnostic studies |
|
2b |
Individual cohort study (or low quality RCT; eg, less than 80% follow-up) |
Retrospective cohort study or follow-up of untreated control patients in an RCT |
Exploratory cohort study with good reference standards |
|
3a |
SR (with homogeneity) of case-control studies |
SR (with homogeneity) of 3b and better studies |
|
|
3b |
Individual case-control study |
Non-consecutive study or without consistently applied reference standards |
|
|
4 |
Case-series (and poor quality cohort and case-control studies) |
Case-series (and poor quality prognostic cohort studies) |
Case-control study, poor or nonindependent reference standard |
|
5 |
Expert opinion without explicit critical appraisal, or based on physiology, bench research or ‘first principles’ |
||
|
Data from reference [26]. RCT Randomized control trial; SR Systematic review |
|||
Can severe hyperbilirubinemia be accurately predicted?
Timed TSB measurements
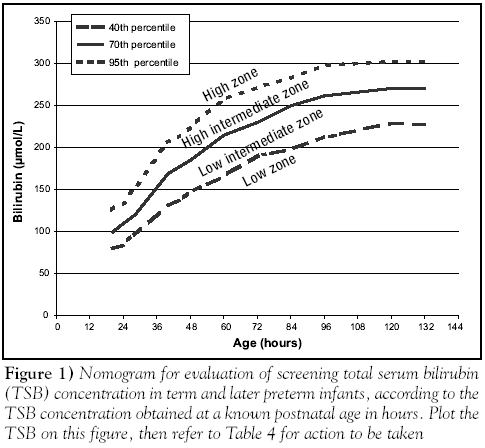
Carefully timed TSB measurements can be used to predict the chances of developing severe hyperbilirubinemia. A study [9] in a North American multiethnic population of appropriate weight for gestational age term and late preterm infants (35 weeks or greater) who did not have a positive direct Coombs test demonstrated that a timed measurement of TSB concentration at discharge (between 18 h and three days of age) could predict a later TSB measurement greater than the 95th percentile within stated confidence limits (the 95th percentile was approximately 300 µmol/L after 96 h of age) (evidence level 1b). When the TSB concentration was below the 40th percentile at the time of measurement, there were no cases of subsequent TSB concentration greater than the 95th percentile. When the TSB concentration was between the 40th and the 75th percentiles, only 2.2% of infants developed a TSB concentration greater than the 95th percentile. Finally, when the TSB concentration was above the 75th percentile, 12.9% of infants subsequently exceeded the 95th percentile [9]. Routine TSB estimation at 6 h of life can also be used in term and late preterm infants to predict a TSB concentration greater than 238 µmol/L in infants with a birth weight of 2 kg to 2.5 kg, and a TSB concentration greater than 289 µmol/L in infants with a birth weight greater than 2.5 kg [15] (evidence level 1b). Combining a timed TSB measurement at younger than 48 h with a clinical risk score improved the prediction of a subsequent TSB concentration greater than 342 µmol/L [27]; this improvement was almost entirely due to the effect of including gestational age (evidence level 2b). Thus, a TSB concentration between the 75th and the 94th percentiles was associated with a 12% risk of subsequent TSB concentration greater than 342 µmol/L in the infant of 36 weeks’ gestation, and with approximately a 3% risk in the infant of 40 weeks’ gestation [28].
Therefore, the best available method for predicting severe hyperbilirubinemia appears to be the use of a timed TSB measurement analyzed in the context of the infant’s gestational age. Infants of less than 38 weeks’ gestation whose TSB concentration is greater than the 75th percentile have a greater than 10% risk of developing severe hyperbilirubinemia; similarly, infants of 39 to 40 weeks’ gestation whose TSB concentration is above the 95th percentile have a greater than 10% risk (evidence level 2b).
Umbilical cord blood TSB
A TSB concentration greater than 30 µmol/L in umbilical cord blood [29] is statistically correlated with a peak neonatal TSB concentration greater than 300 µmol/L, but the positive predictive value is only 4.8% for the term infant, rising to 10.9% in the late preterm infant, and the specificity is very poor (evidence level 1b).
Universal hemoglobin assessment
Although bilirubin is derived from the breakdown of hemoglobin, routine umbilical cord blood hemoglobin or hematocrit measurement does not aid in the prediction of severe hyperbilirubinemia [30] (evidence level 2b).
Blood group and Coombs testing
ABO isoimmunization is a common cause of severe hyperbilirubinemia. Babies whose mothers are blood group O have an OR of 2.9 for severe hyperbilirubinemia (because most infants with jaundice due to ABO isoimmunization are blood group A or B infants born to a mother with group O blood)[31][32]. The need for phototherapy is increased in ABO-incompatible infants who are direct antiglobulin test (DAT [direct Coombs test])-positive compared with those who are DAT-negative [28][30]. Universal testing for incompatibility with blood grouping, and for isoimmunization using the DAT, on cord blood does not improve clinical outcomes compared with testing only infants whose mothers are group O [33][34] (evidence level 2b). Testing all babies whose mothers are group O does not improve outcomes compared with testing only those with clinical jaundice [35][36] (evidence level 2b). Therefore, it is reasonable to perform a DAT in clinically jaundiced infants of mothers who are group O and in infants with an elevated risk of needing therapy (ie, in the high-intermediate zone [Figure 1]). The results will determine whether they are low risk or high risk, and may therefore affect the threshold at which therapy would be indicated (Figure 2).
The usual antenatal screen for a panel of red cell antibodies occasionally identifies additional mothers who will deliver infants at increased risk of hemolysis. The significance of the various antibodies differs; in such infants, analysis of blood group and a DAT is usually required, closer follow-up and earlier therapy may be needed, and a consultation with a paediatric hematologist or neonatologist is suggested.
A full-size downloadable version of this graph
Reproduced and adapted with permission from Pediatrics 2004;114:297-316. © 2004 by the American Academy of Pediatrics.

A full-size downloadable version of this graph
Reproduced and adapted with permission from Pediatrics 2004;114:297-316. © 2004 by the American Academy of Pediatrics.
Glucose-6-phosphate dehydrogenase deficiency
Newborns with glucose-6-phosphate dehydrogenase (G6PD) deficiency have an increased incidence of severe hyperbilirubinemia (evidence level 1b). Testing for G6PD deficiency in babies whose ethnic group or family history suggest an increased risk of G6PD deficiency is advised (eg, Mediterranean , Middle Eastern, African [37] or Southeast Asian origin). Although G6PD deficiency is an X-linked disease, female heterozygotes can have more than 50% of their red cells deficient in the enzyme because of random inactivation of the X chromosome. Females with greater proportions of their red cells affected have an increased risk of severe neonatal hyperbilirubinemia [38]; therefore, testing of both girls and boys who are at risk is advised [39]. G6PD deficiency increases the likelihood of requiring exchange transfusion in infants with severe hyperbilirubinemia; therefore, a test for G6PD deficiency should be considered in all infants with severe hyperbilirubinemia (evidence level 5). It should also be recognized that in the presence of hemolysis, G6PD levels can be overestimated and this may obscure the diagnosis [40]. Females in particular can have misleading results on the common screening tests [41]. G6PD-deficient newborns may require intervention at a lower TSB concentration because they are more likely to progress to severe hyperbilirubinemia [42][43]. Unfortunately, in many centres, it currently takes several days for a G6PD deficiency screening test result to become available. Improving the turnaround time for this test would improve care of the newborn. Because G6PD deficiency is a disease with lifelong implications, testing infants at risk is still of value.
End-tidal carbon monoxide
Exhaled carbon monoxide is increased during hemolysis; however, prediction of severe hyperbilirubinemia is not improved by measuring the end-tidal carbon monoxide concentration [44] in addition to a timed TSB measurement (evidence level 1b).
Recommendations
- All mothers should be tested for ABO and Rh(D) blood types and be screened for red cell antibodies during pregnancy (recommendation grade D [Table 3]).
- If the mother was not tested, cord blood from the infant should be sent for evaluation of the blood group and a DAT (Coombs test) (recommendation grade D).
- Blood group evaluation and a DAT should be performed in infants with early jaundice of mothers of blood group O (recommendation grade B).
- Selected at-risk infants (Mediterranean, Middle Eastern, African or Southeast Asian origin) should be screened for G6PD deficiency (recommendation grade D).
- A test for G6PD deficiency should be considered in all infants with severe hyperbilirubinemia (recommendation grade D).
| TABLE 3 Grades of recommendation |
|
|
A |
Consistent level 1 studies |
|
B |
Consistent level 2 or 3 studies |
|
C |
Level 4 studies |
|
D |
Level 5 evidence or troublingly inconsistent or inconclusive studies of any level |
|
Data from reference [26] |
|
Who should have their bilirubin concentration measured, when and how?
Previous recommendations were to measure TSB concentration in all infants with clinical jaundice at any time in the first four days of life, and to measure TSB concentration in those who are not clinically jaundiced but have increased risk factors. Because of the high occurrence of the risk factors, this recommendation requires TSB measurement in a large majority of infants (exceptions include females of certain ethnic groups who are fully formula fed and more than 37 weeks’ gestation). Despite these recommendations, infants continue to present with severe hyperbilirubinemia during or after their initial hospitalization. Recent data from the CPSP [18] demonstrated that 185 of 289 infants with critical hyperbilirubinemia presented after hospital discharge. There is an opportunity to perform universal screening for either TSB or transcutaneous bilirubin (TcB) before the period of highest risk [19][42] and to use this to determine the risk profile and individualize follow-up. Furthermore, clinical assessment of jaundice is inadequate for diagnosing hyperbilirubinemia. Jaundice is not evident on clinical examination when the TSB concentration is less than 68 µmol/L, and only 50% of babies with a TSB concentration greater than 128 µmol/L appear jaundiced. One study [41] showed a difference of up to 100 µmol/L between visual and laboratory estimates of bilirubin concentration. In one study [43], all infants with a TSB concentration greater than 204 µmol/L were identified as being jaundiced; in another study [45], 19% of infants with TSB concentrations this high were not considered to be jaundiced by neonatologists (evidence level 2b). Although there have been no prospective, controlled trials to evaluate the effectiveness or cost-benefit relationship of universal screening, it appears to be a reasonable strategy, and an observational study [46] has reported it to be effective (evidence level 4).
The peak TSB concentration usually occurs between three and five days of life, at which time the majority of babies have already been discharged from hospital. At the usual age of discharge, TSB concentrations that are in a high-risk zone on the nomograms cannot be reliably detected by visual inspection, especially in infants with darker skin colours. To predict the occurrence of severe hyperbilirubinemia, it is therefore recommended that either TSB or TcB concentration be measured in all infants between 24 h and 72 h of life; if the infant does not require immediate treatment, the results should be plotted on the predictive nomogram to determine the risk of progression to severe hyperbilirubinemia. The TSB (or TcB) concentration and the predictive zone should be recorded, a copy should be given to the family at the time of discharge, and follow-up arrangements should be made for infants who are at higher risk (Table 4).
| TABLE 4 Response to results of bilirubin screening |
|||
|
Zone |
Greater than 37 weeks’ gestation and DAT-negative |
35 to 37 6/7 weeks’ gestation or DAT-positive |
35 to 37 6/7 weeks’ gestation and DAT-positive |
|
High |
Further testing or treatment required* |
Further testing or treatment required* |
Phototherapy required |
|
High-intermediate |
Routine care |
Follow-up within 24 h to 48 |
Further testing or treatment required* |
|
Low-intermediate |
Routine care |
Routine care |
Further testing or treatment required* |
|
Low |
Routine care |
Routine care |
Routine care |
|
*Arrangements must be made for a timely (eg, within 24 h) re-evaluation of bilirubin by serum testing. Depending on the level indicated in Figure 2, treatment with phototherapy may also be indicated. DAT Direct antiglobulin test |
|||
If the TSB concentration had not been measured earlier because of clinical jaundice, a TSB measurement should be obtained at the same time as the metabolic screening test to avoid an increase in the number of painful procedures and to minimize costs; alternatively, a TcB measurement should be obtained either at discharge or before 72 h of life. The prediction of severe hyperbilirubinemia is more accurate if the gestational age at birth is included in the prediction model [28].
Some of the most severely affected infants require therapy to be started before the time of the metabolic screen to prevent severe hyperbilirubinemia and its complications. Sudden increases in TSB concentration may also occasionally occur after the first two to three days [47]. This may occur particularly in association with excessive postnatal weight loss. Therefore, the institution of a program of universal screening complements, but does not replace, careful ongoing assessment of newborn infants beginning from the first hours of life and continuing through the first weeks. Systems to ensure follow-up within the recommended intervals after hospital discharge must be in place so that an infant who develops severe hyperbilirubinemia can be identified and treated promptly. This requires, for example, that an infant discharged from hospital in the first 24 h of life be reviewed within 24 h, any day of the week, by an individual with the training to recognize neonatal hyperbilirubinemia, obtain measurement of TSB or TcB without delay and refer the infant to a treatment facility if required. This individual may be from any medical or nursing discipline.
In addition to universal measurement, all newborns should be clinically assessed for jaundice repeatedly within the first 24 h, and again, at a minimum, 24 h to 48 h later. This should be performed by an individual competent in the assessment of the newborn who can, if necessary, immediately obtain a TSB or TcB measurement and arrange treatment for the infant, whether in hospital or after discharge.
Measurement of bilirubin
It is possible to measure bilirubin concentration using capillary or venous blood samples or transcutaneously. There is no systematic difference between the results of capillary or venous samples [48][49]. Capillary sampling is the method used most often in Canada and in most studies, including those of Bhutani et al [9]. There are several limitations to TcB measurements [50]: they become unreliable after initiation of phototherapy [51], and they may be unreliable with changes in skin colour and thickness [52]. However, the results are more accurate at lower levels of bilirubin, and therefore, use of TcB as a screening device is reasonable [46]. The available devices differ in accuracy; safe use of the device mandates knowledge of the accuracy of the particular device. The 95% CIs for TSB concentration based on the TcB measurement range from approximately 37 µmol/L to 78 µmol/L [50][53]. For example, if the 95% CI is 37 µmol/L, then a TcB concentration greater than 37 µmol/L below the treatment threshold on Figure 2 should be safe (ie, if the threshold at 24 h is 170 µmol/L, then a TcB of less than 133 µmol/L should be safe) (evidence level 1b).
Measurement of free bilirubin
Displacement of bilirubin from albumin-binding sites by certain toxic medications and additives has caused numerous cases of kernicterus in the past, mostly in the neonatal intensive care unit population [54]. It is believed to be free bilirubin (ie, not bound to albumin) that crosses the blood-brain barrier and causes neuronal damage [55]-[57]. The clinical value of measurement of free bilirubin is currently uncertain and it is not readily available [58].
Measurement of conjugated bilirubin
Although early neonatal jaundice is generally due to unconjugated hyperbilirubinemia, in some situations the conjugated fraction may be elevated, such as in rhesus erythroblastosis, liver disease and cholestasis [59]. In infants placed on phototherapy, measurement of the conjugated fraction should be considered. However, previous reports [16][20] on the epidemiology of bilirubin toxicity use the TSB concentration as the standard, which remains the deciding value for phototherapy and other therapies. The conjugated bilirubin fraction should be estimated in an infant with persistent jaundice (longer than two weeks) and/or hepatosplenomegaly [60]. A total conjugated bilirubin concentration greater than 18 µmol/L or greater than 20% of the TSB concentration warrants further investigation [61][62].
Recommendations
- Either TSB or TcB concentration should be measured in all infants during the first 72 h of life. If not required earlier because of clinical jaundice, a TSB measurement should be obtained at the same time as the metabolic screening test; alternatively, a TcB measurement should be obtained either at discharge or, if not yet discharged, at 72 h of life (recommendation grade C).
- If the TSB concentration does not require immediate intervention, the results should be plotted on the predictive nomogram. The result of the TSB measurement, the time at which it was obtained and the zone should be recorded, and a copy should be given to the parents. Follow-up of the infant should be individualized according to the risk assessment (recommendation grade D).
- Any infant discharged before 24 h of life should be reviewed within 24 h by an individual with experience in the care of the newborn who has access to testing and treatment facilities (recommendation grade D).
- There should be a systematic approach to the risk assessment of all infants before discharge and institution of follow-up care if the infant develops jaundice (recommendation grade D).
- All newborns who are visibly jaundiced in the first 24 h of life should have their bilirubin level determined (recommendation grade D).
- Transcutaneous bilirubinometry is an acceptable method, either as a routine procedure or in infants with visible jaundice. The result should be summed with the 95% CI of the device to estimate the maximum probable TSB concentration (recommendation grade C).
- TSB concentration may be estimated on either a capillary or a venous blood sample (recommendation grade C).
- Infants with severe or prolonged hyperbilirubinemia should be further investigated, including measurement of the conjugated component of bilirubin (recommendation grade C).
How can the risk of severe hyperbilirubinemia be reduced?
Primary prevention of severe hyperbilirubinemia
Breastfeeding support
Although breastfed infants are at a higher risk for developing severe hyperbilirubinemia than are formula-fed infants, the known risks of acute bilirubin encephalopathy are very small when weighed against the substantial known benefits of breastfeeding [17][63]. Support of the breastfeeding mother by knowledgeable individuals increases the frequency and duration of breastfeeding. It is difficult to find reliable evidence that the risk of severe jaundice can be minimized by a program of breastfeeding support, but other aspects of breastfeeding difficulty can be reduced by such programs, and providing such support is reasonable (evidence level 5) [60]. Exclusively breastfed infants experience their maximum weight loss by day 3 and lose, on average, 6% to 8% of their birth weight [64]. Infants who lose more than 10% of their birth weight should be carefully evaluated by an individual with training and experience in support of breastfeeding mothers [64][65] (evidence level 5). Routine supplementation of breastfed infants with water or dextrose water does not appear to prevent hyperbilirubinemia (evidence level 2b) [66].
Other ineffective interventions
Routine use of glycerine suppositories [64][67], routine glycerine enemas [65], L-aspartic acid, enzymatically hydrolyzed casein, whey/casein and clofibrate [68] have all been studied in small randomized controlled trials (RCTs), but their use has been found to have no effect on clinically important outcomes.
Prevention of severe hyperbilirubinemia in infants with hemolysis
Phenobarbitone
Phenobarbitone, studied as a means of preventing severe hyperbilirubinemia in infants with G6PD deficiency [69], did not improve clinically important outcomes (evidence level 1b).
Tin-mesoporphyrin
Synthetic analogues of heme oxygenase, such as tin-mesoporphyrin (SnMP), strongly inhibit its activity and suppress the production of bilirubin. In a study [70] with historical controls in infants with G6PD deficiency, SnMP eliminated the need for phototherapy and appeared to prevent severe hyperbilirubinemia. However, prospective RCTs have as yet failed to demonstrate a clinically important benefit (evidence level 1b), and the compounds are not commercially available [71][72].
Prophylactic phototherapy
A quasi-RCT (73; n=142) was unable to find clinical benefit of prophylactic phototherapy in ABO isoimmunization (evidence level 2b).
Prevention of severe hyperbilirubinemia in infants with mild or moderate hyperbilirubinemia
Phototherapy
Phototherapy can be used both to prevent severe hyperbilirubinemia in infants with a moderately elevated TSB concentration and as initial therapy in those with severe hyperbilirubinemia.
The energy from light induces a conformational change in the bilirubin molecule, making it water soluble; light in the blue-green part of the spectrum is most effective. The effectiveness of phototherapy is related to the area of skin exposed and the intensity of the light at the skin at the relevant wavelengths [74]-[76]. More intense phototherapy can be achieved using multiple phototherapy units [77] or simply moving the unit closer to the infant. Although phototherapy increases water loss from transepidermal skin, this is not a clinically important issue in full-term infants who are drinking well. Side effects of phototherapy include temperature instability, intestinal hypermotility, diarrhea, interference with maternal-infant interaction and, rarely, bronze discolouration of the skin [41]. Phototherapy in the neonatal period is perceived by parents as implying that their infant’s jaundice was a serious disease [78], and is associated with increased anxiety and health care use (evidence level 2). Reassurance of the parents that appropriate intervention and follow-up will prevent any consequences of hyperbilirubinemia is an important part of the care of these infants. Eye patches should be used to protect the developing retina because animal studies demonstrate a potential risk [79].
Phototherapy decreases the progression to severe hyperbilirubinemia in infants with moderate hyperbilirubinemia (evidence level 1a) [12]. Some infants with jaundice are dehydrated, and rehydration will usually lead to a prompt fall in the TSB concentration; enteral feeding should be continued because it will replace missing fluid, supply energy and reduce enterohepatic reuptake of the bilirubin [80].
In general, fluorescent light is most commonly used [81]; the intensity of light produced by fluorescent tubes wanes over time. A program of biomedical support for ensuring adequate light intensity is important to assure effective therapy. Fibre optic phototherapy systems were introduced in the late 1980s; the advantages are that the baby can be breastfed without interruption of phototherapy and eye pads are not required, but the disadvantage is that the peak intensity is less than that of fluorescent systems. Halogen spotlights may also be used, but they must not be placed closer to the infant than the manufacturer’s recommendations.
Intensive phototherapy, as recommended by the present position statement, implies that a high intensity of light (greater than 30 µW/cm2/nm) is applied to the greatest surface area of the infant possible. In usual clinical situations, this will require two phototherapy units, or special high-intensity fluorescent tubes, placed approximately 10 cm from the infant, who can be nursed in a bassinet. Usually the diaper can be left in place. In infants whose TSB concentration is approaching the exchange transfusion threshold, the addition of a fibre optic blanket under the infant can increase the surface area illuminated, and the diaper should then be removed (or a phototherapy wavelength-transmitting diaper used instead). The guidelines for therapy (Figure 2) are based on limited direct evidence, but the Canadian Paediatric Society’s Fetus and Newborn Committee believed that the consensus of the American Academy of Pediatrics’ Subcommittee on Hyperbilirubinemia was the most appropriate currently available standard [20]. Conventional phototherapy – a single bank of fluorescent lights placed above the incubator of an infant nursed with a diaper in place – is, of course, less effective because both surface area and intensity are reduced; nevertheless, it will have an effect on TSB concentration.
The recommendations for treatment are determined from Figure 2. These recommendations are as follows:
- Intensive phototherapy for infants with severe hyperbilirubinemia or those at greatly elevated risk of developing severe hyperbilirubinemia.
- In addition, there is an option for conventional phototherapy for infants with a moderately elevated risk and a TSB concentration of 35 µmol/L to 50 µmol/L below the thresholds on Figure 2.
A useful online tool is available for deciding whether intensive phototherapy would be recommended by these guidelines [82].
Interrupting breastfeeding
Interrupting breastfeeding as part of therapy for hyperbilirubinemia is associated with a major increase in the frequency of stopping breastfeeding by one month (RR=1.79, 95% CI 1.04 to 3.06, number needed to harm = 4 [evidence level 2b]) [83]. Continued breastfeeding in jaundiced infants receiving phototherapy is not associated with adverse clinical outcomes, although an observational study [84] showed a slower response to phototherapy in the first 24 h in exclusively breastfed infants compared with those who received supplementation (bilirubin decreases of 17.1% versus 22.9%, respectively; P=0.03). The duration of phototherapy did not differ between the groups, and no other clinically important outcomes were affected. An RCT [85] in jaundiced breastfed newborns showed no clinically significant difference in the frequency of TSB concentration reduction to normal concentration at 48 h if breastfeeding was interrupted, either in groups undergoing phototherapy (RR=1.07, 95% CI 0.6 to 1.92; P=0.818) or in those who did not have phototherapy. There were no clinically important differences in outcomes.
Intravenous immunoglobulin
Intravenous immunoglobulin (IVIG) reduces bilirubin concentrations in newborns with rhesus hemolytic disease and other immune hemolytic jaundice. It acts as a completive inhibitor for those antibodies that cause red cell destruction, release hemoglobin and cause jaundice [47]. A systematic review of three prior RCTs [86] and a subsequent RCT [87] demonstrated a significant reduction in the need for exchange transfusion in infants with significant jaundice randomly assigned to receive IVIG at a dose of either 500 mg/kg or 1 g/kg (from the Cochrane review, RR=0.28, 95% CI 0.17 to 0.47, number needed to treat = 3 [evidence level 1a]). The entry criteria for each of these studies differed, making exact treatment indications difficult to determine. It appears reasonable to initiate this treatment in infants with predicted severe disease based on antenatal investigation and in those with an elevated risk of needing exchange transfusion based on the postnatal progression of TSB concentration.
SnMP
SnMP, studied for preventing the progression of moderate hyperbilirubinemia [88], showed no evidence of reduction in clinically important outcomes (evidence level 1a).
Supplemental fluids
Infants with nonhemolytic jaundice, not obviously dehydrated, with a TSB concentration between 308 µmol/L and 427 µmol/L were randomly assigned to either a control group or to a group receiving extra fluids (by intravenous infusion, then orally) in a small RCT from northern India [89]. The frequency of exchange transfusion was significantly reduced by the extra fluids from 54% to 16% [89] (evidence level 1b). Oral fluids appear to be as effective as intravenous fluids [90] during intensive phototherapy (evidence level 1b).
There is observational evidence that offering supplemental oral fluids may interfere with the eventual duration of breastfeeding [78] (evidence level 2b), but such studies were not performed in the context of brief supplementation in the setting of neonatal jaundice, and a systematic review of intervention studies found no reliable evidence [91]. The frequency of exchange transfusion in the infants in the study noted above was very high [92]; the same absolute risk reduction from extra fluids will not be seen in a population with a much lower likelihood of requiring exchange. Therefore, in breastfed infants, extra fluids are indicated for, but should be restricted to, infants with an elevated risk of requiring exchange transfusion (evidence level 1b).
Agar
Oral agar to prevent enterohepatic reuptake of bilirubin is not supported by the available evidence [92]-[95] (evidence level 1b).
Recommendations
- A program for breastfeeding support should be instituted in every facility where babies are delivered (recommendation grade D).
- Routine supplementation of breastfed infants with water or dextrose water is not recommended (recommendation grade B).
- Infants with a positive DAT who have predicted severe disease based on antenatal investigation or an elevated risk of progressing to exchange transfusion based on the postnatal progression of TSB concentration should receive IVIG at a dose of 1 g/kg (recommendation grade A).
- A TSB concentration consistent with increased risk (Figure 1) and (Table 4) should lead to enhanced surveillance for development of severe hyperbilirubinemia, with follow-up within 24 h to 48 h, either in hospital or in the community, and repeat estimation of TSB or TcB concentration in most circumstances (recommendation grade C).
- Intensive phototherapy should be given according to the guidelines shown in Figure 2 (recommendation grade D).
- Conventional phototherapy is an option at TSB concentrations 35 µmol/L to 50 µmol/L lower than the guidelines in Figure 2) (recommendation grade D).
- Breastfeeding should be continued during phototherapy (recommendation grade A).
- Supplemental fluids should be administered, orally or by intravenous infusion, in infants receiving phototherapy who are at an elevated risk of progressing to exchange transfusion (recommendation grade A).
How should severe hyperbilirubinemia be treated?
Phototherapy
An infant who presents with severe hyperbilirubinemia, or who progresses to severe hyperbilirubinemia despite initial treatment, should receive immediate intensive phototherapy. The bilirubin concentration should be checked within 2 h to 6 h of initiation of treatment to confirm response. Consideration of further therapy should commence and preparations for exchange transfusion may be indicated. Supplemental fluids are indicated, and IVIG should be given if not already commenced for the infant with isoimmunization.
Exchange transfusion
If phototherapy fails to control the rising bilirubin concentrations, exchange transfusion is indicated to lower TSB concentrations. For healthy term newborns without risk factors, exchange transfusion should be considered when the TSB concentration is between 375 µmol/L and 425 µmol/L (despite adequate intensive phototherapy). Because blood collected after an exchange transfusion is of no value for investigating many of the rarer causes of severe hyperbilirubinemia, these investigations should be considered before performing the exchange transfusion. Appropriate amounts of blood should be taken and stored for tests such as those for red cell fragility, enzyme deficiency (G6PD or pyruvate kinase deficiency) and metabolic disorders, as well as for hemoglobin electrophoresis and chromosome analysis. Preparation of blood for exchange transfusion may take several hours, during which time intensive phototherapy, supplemental fluids and IVIG (in case of isoimmunization) should be used. If an infant whose TSB concentration is already above the exchange transfusion line presents for medical care, then repeat measurement of the TSB concentration just before performance of the exchange is reasonable, as long as therapy is not thereby delayed. In this way, some exchange transfusions, with their attendant risks, may be avoided. Exchange transfusion is a procedure with substantial morbidity that should only be performed in centres with the appropriate expertise under supervision of an experienced neonatologist. An infant with clinical signs of acute bilirubin encephalopathy should have an immediate exchange transfusion (evidence level 4).
Recommendations
- Infants with a TSB concentration above the thresholds shown on Figure 3 should have immediate intensive phototherapy, and should be referred for further investigation and preparation for exchange transfusion (recommendation grade B).
- An infant with clinical signs of acute bilirubin encephalopathy should have an immediate exchange transfusion (recommendation grade D).

For a full-size downloadable version of this graph.
Reproduced and adapted with permission from Pediatrics 2004;114:297-316. © 2004 by the American Academy of Pediatrics.
Follow-up
Routine newborn surveillance, whether in hospital or after discharge, should include assessment of breastfeeding and jaundice every 24 h to 48 h until feeding is established (usually on the third or fourth day of life). All jaundiced infants, especially high-risk infants and those who are exclusively breastfed, should continue to be closely monitored until feeding and weight gain are established and the TSB concentration starts to fall. Community services should include breastfeeding support and access to TSB or TcB testing. Infants with isoimmunization are at risk for severe anemia after several weeks; it is suggested that a repeat hemoglobin measurement be performed at two weeks if it was low at discharge and at four weeks if it was normal (evidence level 5). Infants requiring exchange transfusion or those who exhibit neurological abnormalities should be referred to regional multidisciplinary follow-up programs. Neurosensory hearing loss is of particular importance in infants with severe hyperbilirubinemia, and their hearing screen should include brainstem auditory evoked potentials.
Further investigations
The occurrence of severe hyperbilirubinemia mandates an investigation of the cause of hyperbilirubinemia. Investigations should include a clinically pertinent history of the baby and the mother, family history, description of the labour and delivery, and the infant’s clinical course [35]. A physical examination should be supplemented by laboratory investigations (conjugated and unconjugated bilirubin levels; direct Coombs test; hemoglobin and hematocrit levels; and complete blood cell count, including differential count, blood smear and red cell morphology). Investigations for sepsis should be performed if warranted by the clinical situation.
Recommendations
- Adequate follow-up should be ensured for all infants who are jaundiced (recommendation grade D).
- Infants requiring intensive phototherapy should be investigated for determination of the cause of jaundice (recommendation grade C).
Conclusion
Severe hyperbilirubinemia in relatively healthy term or late preterm newborns (greater than 35 weeks’ gestation) continues to carry the potential for complications from acute bilirubin encephalopathy and chronic sequelae. Careful assessment of the risk factors involved, a systematic approach to the detection and follow-up of jaundice with the appropriate laboratory investigations, along with judicious phototherapy and exchange transfusion when indicated, are all essential to avoid these complications.
Acknowledgements
This position statement was reviewed by the Canadian Paediatric Society’s Community Paediatrics Committee and the College of Family Physicians of Canada.
FETUS AND NEWBORN COMMITTEE
Members: Khalid Aziz MD (board representative 2000-2006); Keith J Barrington MD (chair); Joanne Embree MD (board representative); Haresh Kirpalani MD; Koravangattu Sankaran MD; Hilary Whyte MD (sabbatical 2006-2007); Robin Whyte MD
Liaisons: Dan Farine MD, Society of Obstetricians and Gynaecologists of Canada; David Keegan MD, College of Family Physicians of Canada, Maternity and Newborn Care Committee; Francine Lefebvre MD, Neonatal-Perinatal Medicine Section, Canadian Paediatric Society; Catherine McCourt MD, Health Surveillance and Epidemiology, Public Health Agency of Canada; Shahirose Premji, Neonatal Nurses; Alfonso Solimano MD, Neonatal-Perinatal Medicine Section, Canadian Paediatric Society (2002–2006); Ann Stark MD, Committee on Fetus and Newborn, American Academy of Pediatrics; Amanda Symington, Neonatal Nurses (1999–2006)
Principal authors: Keith J Barrington MD; Koravangattu Sankaran MD
References
- Hansen TW. Mechanisms of bilirubin toxicity: Clinical implications. Clin Perinatol 2002;29:765-778,viii.
- Newman TB, Maisels MJ. Evaluation and treatment of jaundice in the term newborn: A kinder, gentler approach. Pediatrics 1992;89(5 Pt 1):809-18.
- Volpe JJ. Neurology of the Newborn, 4th Edition. Philadelphia : WB Saunders, 2001.
- American Academy of Pediatrics, Subcommittee on Neonatal Hyperbilirubinemia. Neonatal jaundice and kernicterus. Pediatrics 2001;108:763-5.
- Penn AA, Enzmann DR, Hahn JS, Stevenson DF. Kernicterus in a full term infant. Pediatrics 1994;93(6 Pt 1):1003-6.
- Maisels MJ, Newman TB. Kernicterus in otherwise healthy, breast-fed term newborns. Pediatrics 1995;96(4 Pt 1):730-3.
- Stevenson DK, Fanaroff AA, Maisels MJ, et al. Prediction of hyperbilirubinemia in near-term and term infants. Pediatrics 2001;108:31-9.
- Harris MC, Bernbaum JC, Polin JR, Zimmerman R, Polin RA. Developmental follow-up of breastfed term and near-term infants with marked hyperbilirubinemia. Pediatrics 2001;107:1075-80.
- Bhutani VK, Johnson L, Sivieri EM. Predictive ability of a predischarge hour-specific serum bilirubin for subsequent significant hyperbilirubinemia in healthy term and near-term newborns. Pediatrics 1999;103:6-14.
- Bhutani VK, Johnson LH, Maisels JM, et al. Kernicterus: Epidemiological strategies for its prevention through systems-based approaches. J Perinatol 2004;24:650-62.
- American Academy of Pediatrics, Subcommittee on Neonatal Hyperbilirubinemia. Management of hyperbilirubinemia in the newborn infant 35 or more weeks of gestation. Pediatrics 2004;114:297-316. (Erratum in 2004;114:1138).
- Ip S, Glicken S, Kulig J, et al. Management of Neonatal Hyperbilirubinemia. Evidence Report/Technology Assessment No. 65 (Prepared by Tufts-New England Medical Center Evidence-based Practice Center under Contract No. 290-97-0019). AHRQ Publication No. 03-E011. Rockville, MD: US Department of Health and Human Services, Agency for Healthcare Research and Quality, 2003.
- Ohlsson A. Neonatal jaundice: Continuing concern and need for research. Pediatr Res 2001;50:674-5.
- Hintz SR, Stevenson DK. Just when you thought it was safe. Pediatr Res 2001;50:676-7.
- Sarici SU, Serdar MA, Korkmaz A, et al. Incidence, course, and prediction of hyperbilirubinemia in near-term and term newborns. Pediatrics 2004;113:775-80.
- Ip S, Chung M, Kulig J, et al; American Academy of Pediatrics, Subcommittee on Hyperbilirubinemia. An evidence-based review of important issues concerning neonatal hyperbilirubinemia. Pediatrics 2004;114:e130-53.
- Johnson LH, Bhutani VK, Brown AK. System-based approach to management of neonatal jaundice and prevention of kernicterus. J Pediatr 2002;140:396-403.
- Sgro M, Campbell D, Shah V. Incidence and causes of severe neonatal hyperbilirubinemia in Canada. CMAJ 2006;175:587-90.
- Centers for Disease Control and Prevention (CDC). Kernicterus in full term infants – United States, 1994-1998. MMWR Morb Mortal Wkly Rep 2001;50:491-4.
- Maisels MJ, Newman TB. Jaundice in full-term and near-term babies who leave the hospital within 36 hours. The pediatrician’s nemesis. Clin Perinatol 1998;25:295-302.
- Ebbesen F. Recurrence of kernicterus in term and near-term infants in Denmark. Acta Paediatr 2000;89:1213-7.
- Dennery PA, Seidman DS, Stevenson DK. Neonatal hyperbilirubinemia. N Engl J Med 2001;344:581-90.
- Soorani-Lunsing I, Woltil HA, Hadders-Algra M. Are moderate degrees of hyperbilirubinemia in healthy term neonates really safe for the brain? Pediatr Res 2001;50:701-5.
- Seidman DS, Ergaz Z, Paz I, et al. Predicting the risk of jaundice in full-term healthy newborns: A prospective population-based study. J Perinatol 1999;19(8 Pt 1):564-7.
- Haynes RB, McKibbon KA, Wilczynski NL, Walter SD, Werre SR; Hedges Team. Optimal search strategies for retrieving scientifically strong studies of treatment from Medline: Analytical survey. BMJ 2005;330:1179.
- Oxford Centre for Evidence-Based Medicine. Version current at March 28, 2007.
- Newman TB, Liljestrand P, Escobar GJ. Combining clinical risk factors with serum bilirubin levels to predict hyperbilirubinemia in newborns. Arch Pediatr Adolesc Med 2005;159:113-9.
- Sarici SU, Yurdakok M, Serdar MA, et al. An early (sixth-hour) serum bilirubin measurement is useful in predicting the development of significant hyperbilirubinemia and severe ABO hemolytic disease in a selective high-risk population of newborns with ABO incompatibility. Pediatrics 2002;109:e53.
- Carbonell X, Botet F, Figueras J, Riu-Godo A. Prediction of hyperbilirubinaemia in the healthy term newborn. Acta Paediatr 2001;90:166-70.
- Madlon-Kay DJ. The clinical significance of ABO blood group incompatibility. Arch Fam Med 1993;2:285-7.
- Ozolek JA, Watchko JF, Mimouni F. Prevalence and lack of clinical significance of blood group incompatibility in mothers with blood type A or B. J Pediatr 1994;125:87-91.
- Newman TB, Easterling MJ, Goldman ES, Stevenson DK. Laboratory evaluation of jaundice in newborns. Frequency, cost, and yield. Am J Dis Child 1990;144:364-8. (Erratum in 1992;146:1420-1)
- Dinesh D. Review of positive direct antiglobulin tests found on cord blood sampling. J Paediatr Child Health 2005;41:504-7.
- Leistikow EA, Collin MF, Savastano GD, de Sierra TM, Leistikow BN. Wasted health care dollars. Routine cord blood type and Coombs’ testing. Arch Pediatr Adolesc Med 1995;149:1147-51.
- Madan A, Huntsinger K, Burgos A, Benitz WE. Readmission for newborn jaundice: The value of the Coombs’ test in predicting the need for phototherapy. Clin Pediatr (Phila) 2004;43:63-8.
- Madlon-Kay DJ. Identifying ABO incompatibility in newborns: Selective vs automatic testing. J Fam Pract 1992;35:278-80.
- Kaplan M, Herschel M, Hammerman C, Karrison T, Hoyer JD, Stevenson DK. Studies in hemolysis in glucose-6-phosphate dehydrogenase-deficient African American neonates. Clin Chim Acta 2006;365:177-82.
- Meloni T, Forteleoni G, Dore A, Cutillo S. Neonatal hyperbilirubinaemia in heterozygous glucose-6-phosphate dehydrogenase deficient females. Br J Haematol 1983;53:241-6.
- Kaplan M, Beutler E, Vreman HJ, et al. Neonatal hyperbilirubinemia in glucose-6-phosphate dehydrogenase-deficient heterozygotes. Pediatrics 1999;104(1 Pt 1):68-74.
- Gourley GR, Kreamer B, Arend R. The effect of diet on feces and jaundice during the first 3 weeks of life. Gastroenterology 1992;103:660-7.
- Riskin A, Kugelman A, Abend-Weinger M, Green M, Hemo M, Bader D. In the eye of the beholder: How accurate is clinical estimation of jaundice in newborns? Acta Paediatr 2003;92:574-6. (Erratum in 2005;94:1168)
- Bhutani VK, Gourley GR, Adler S, Kreamer B, Dalin C, Johnson LH. Noninvasive measurement of total serum bilirubin in a multiracial predischarge newborn population to assess the risk of severe hyperbilirubinemia. Pediatrics 2000;106:e17.
- Madlon-Kay DJ. Recognition of the presence and severity of newborn jaundice by parents, nurses, physicians, and icterometer. Pediatrics 1997;100:E3.
- Stevenson DK, Fanaroff AA, Maisels MJ, et al. Prediction of hyperbilirubinemia in near-term and term infants. J Perinatol 2001;21(Suppl 1):S63-72.
- Riskin A, Abend-Weinger M, Bader D. How accurate are neonatologists in identifying clinical jaundice in newborns? Clin Pediatr (Phila) 2003;42:153-8.
- Eggert LD, Wiedmeier SE, Wilson J, Christensen RD. The effect of instituting a prehospital-discharge newborn bilirubin screening program in an 18-hospital health system. Pediatrics 2006;117:e855-62.
- Watchko JF. Vigintiphobia revisited. Pediatrics 2005;115:1747-53.
- Leslie GI, Philips JB III, Cassady G. Capillary and venous bilirubin values. Are they really different? Am J Dis Child 1987;141:1199-200.
- Amato M, Huppi P, Markus D. Assessment of neonatal jaundice in low birth weight infants comparing transcutaneous, capillary and arterial bilirubin levels. Eur J Pediatr 1990;150:59-61.
- Rubaltelli FF, Gourley GR, Loskamp N, et al. Transcutaneous bilirubin measurement: A multicentre evaluation of a new device. Pediatrics 2001;107:1264-71.
- Cremer RJ, Perryman PW, Richards DH. Influence of light on the hyperbilirubinaemia of infants. Lancet 1958;1:1094-7.
- Rubaltelli FF, Carli M. The effect of light on cutaneous bilirubin. Biol Neonate 1971;18:457-62.
- Maisels MJ, Ostrea EM Jr, Touch S, et al. Evaluation of a new transcutaneous bilirubinometer. Pediatrics 2004;113:1628-35.
- Hansen TW. Recent advances in the pharmacotherapy for hyperbilirubinaemia in the neonate. Expert Opin Pharmacother 2003;4:1939-48.
- Ahlfors CE. Unbound bilirubin associated with kernicterus: A historical approach. J Pediatr 2000;137:540-4.
- Funato M, Tamai H, Shimada S, Nakamura H. Vigintiphobia, unbound bilirubin, and auditory brainstem responses. Pediatrics 1994;93:50-3.
- Nakamura H, Yonetani M, Uetani Y, Funato M, Lee Y. Determination of serum unbound bilirubin for prediction of kernicterus in low birthweight infants. Acta Paediatr Jpn 1992;34:642-7.
- McDonagh AF, Maisels MJ. Bilirubin unbound: Deja vu all over again? Pediatrics 2006;117:523-5.
- Venigalla S, Gourley GR. Neonatal cholestasis. Semin Perinatol 2004;28:348-55.
- Butler DA, MacMillan JP. Relationship of breast feeding and weight loss to jaundice in the newborn period: Review of the literature and results of a study. Cleve Clin Q 1983;50:263-8.
- Keffler S, Kelly DA, Powell JE, Green A. Population screening for neonatal liver disease: A feasibility study. J Pediatr Gastroenterol Nutr 1998;27:306-11.
- Powell JE, Keffler S, Kelly DA, Green A. Population screening for neonatal liver disease: Potential for a community-based programme. J Med Screen 2003;10:112-6.
- Maisels MJ, Gifford K. Breast-feeding, weight loss, and jaundice. J Pediatr 1983;102:117-8.
- Bader D, Yanir Y, Kugelman A, Wilhelm-Kafil M, Riskin A. Induction of early meconium evacuation: Is it effective in reducing the level of neonatal hyperbilirubinemia? Am J Perinatol 2005;22:329-33.
- Chen JY, Ling UP, Chen JH. Early meconium evacuation: Effect on neonatal hyperbilirubinemia. Am J Perinatol 1995;12:232-4.
- Nicoll A, Ginsburg R, Tripp JH. Supplementary feeding and jaundice in newborns. Acta Pediatr Scand 1982;71:759-61.
- Perez EM, Cooper TR, Moise AA, Ferry GD, Weisman LE. Treatment of obstructive jaundice in erythroblastosis fetalis with ursodeoxycholic acid (UDCA): A case report. J Perinatol 1998;18:317-9.
- Lindenbaum A, Hernandorena X, Vial M, et al. [Clofibrate for the treatment of hyperbilirubinemia in neonates born at term: A double blind controlled study (author’s transl)]. Arch Fr Pediatr 1981;38(Suppl 1):867-73.
- Murki S, Dutta S, Narang A, Sarkar U, Garewal G. A randomized, triple-blind, placebo-controlled trial of prophylactic oral phenobarbital to reduce the need for phototherapy in G6PD-deficient neonates. J Perinatol 2005;25:325-30.
- Kappas A, Drummond GS, Valaes T. A single dose of Sn-mesoporphyrin prevents development of severe hyperbilirubinemia in glucose-6-phosphate dehydrogenase-deficient newborns. Pediatrics 2001;108:25-30.
- Valaes T, Drummond GS, Kappas A. Control of hyperbilirubinemia in glucose-6-phosphate dehydrogenase-deficient newborns using an inhibitor of bilirubin production, Sn-mesoporphyrin. Pediatrics 1998;101:E1.
- Kappas A, Drummond GS, Manola T, Petmezaki S, Valaes T. Sn-protoporphyrin use in the management of hyperbilirubinemia in term newborns with direct Coombs-positive ABO incompatibility. Pediatrics 1988;81:485-97.
- Yaseen H, Khalaf M, Rashid N, Darwich M. Does prophylactic phototherapy prevent hyperbilirubinemia in neonates with ABO incompatibility and positive Coombs’ test? J Perinatol 2005;25:590-4.
- Warshaw JB, Gagliardi J, Patel A. A comparison of fluorescent and nonfluorescent light sources for phototherapy. Pediatrics 1980;65:795-8.
- Vogl TP. Phototherapy of neonatal hyperbilirubinemia: Bilirubin in unexposed areas of the skin. J Pediatr 1974;85:707-10.
- Seidman DS, Moise J, Ergaz Z, et al. A new blue light-emitting phototherapy device: A prospective randomized controlled study. J Pediatr 2000;136:771-4.
- Sisson TR, Kendall N, Shaw E, Kechavarz-Oliai L. Phototherapy of jaundice in the newborn infant. II. Effect of various light intensities. J Pediatr 1972:81:35-8.
- Casiday RE, Wright CM, Panter-Brick C, Parkinson KN. Do early infant feeding patterns relate to breast-feeding continuation and weight gain? Data from a longitudinal cohort study. Eur J Clin Nutr 2004;58:1290-6.
- Messner KH, Maisels MJ, Leure-DuPree AE. Phototoxicity to the newborn primate retina. Invest Ophthalmol Vis Sci 1978;17:178-82.
- Brown AK, Kim MH, Wu PY, Bryla DA. Efficacy of phototherapy in prevention and management of neonatal hyperbilirubinemia. Pediatrics 1985;75(2 Pt 2):393-400.
- Mills JF, Tudehope D. Fibreoptic phototherapy for neonatal jaundice. Cochrane Database Syst Rev 2001;(1):CD002060.
- BiliTool. http://bilitool.org (Version current at March 5, 2007 ).
- Kemper K, Forsyth B, McCarthy P. Jaundice, terminating breast-feeding, and the vulnerable child. Pediatrics 1989;84:773-8.
- Tan KL. Decreased response to phototherapy for neonatal jaundice in breast-fed infants. Arch Pediatr Adolesc Med 1998;152:1187-90.
- Martinez JC, Maisels MJ, Otheguy L, et al. Hyperbilirubinemia in the breast-fed newborn: A controlled trial of four interventions. Pediatrics 1993;91:470-3.
- Alcock GS, Liley H. Immunoglobulin infusion for isoimmune haemolytic jaundice in neonates. Cochrane Database Syst Rev 2002;(3):CD003313.
- Miqdad AM, Abdelbasit OB, Shaheed MM, Seidahmed MZ, Abomelha AM, Arcala OP. Intravenous immunoglobulin G (IVIG) therapy for significant hyperbilirubinemia in ABO hemolytic disease of the newborn. J Matern Fetal Neonatal Med 2004;16:163-6.
- Suresh GK, Martin CL, Soll RF. Metalloporphyrins for treatment of unconjugated hyperbilirubinemia in neonates. Cochrane Database Syst Rev 2003;(2):CD004207.
- Mehta S, Kumar P, Narang A. A randomized controlled trial of fluid supplementation in term neonates with severe hyperbilirubinemia. J Pediatr 2005;147:781-5.
- Boo NY, Lee HT. Randomized controlled trial of oral versus intravenous fluid supplementation on serum bilirubin level during phototherapy of term infants with severe hyperbilirubinaemia. J Paediatr Child Health 2002;38:151-5.
- Horvath A, Koletzko B, Kalisz M, Szajewska H. The effect of supplemental fluids or feedings during the first days of life on the success and duration of breastfeeding: A systematic review of randomized controlled trials. Arch Pediatr Adolesc Med 2005;159:597-8.
- Odell GB, Gutcher GR, Whitington PF, Yang G. Enteral administration of agar as an effective adjunct to phototherapy of neonatal hyperbilirubinemia. Pediatr Res 1983;17:810-4.
- Kemper K, Horwitz RI , McCarthy P. Decreased neonatal serum bilirubin with plain agar: A meta-analysis. Pediatrics 1988;82:631-8.
- Caglayan S, Candemir H, Aksit S, Kansoy S, Asik S, Yaprak I. Superiority of oral agar and phototherapy combination in the treatment of neonatal hyperbilirubinemia. Pediatrics 1993;92:86-9.
- Ebbesen F, Moller J. Agar ingestion combined with phototherapy in jaundiced newborn infants. Biol Neonate 1977;31:7-9.
Disclaimer: The recommendations in this position statement do not indicate an exclusive course of treatment or procedure to be followed. Variations, taking into account individual circumstances, may be appropriate. Internet addresses are current at time of publication.
Last updated: Feb 2, 2021
